 Calyculin A 11,13,21-triacetate (12) BDBM40779
Calyculin A 11,13,21-triacetate (12) BDBM40779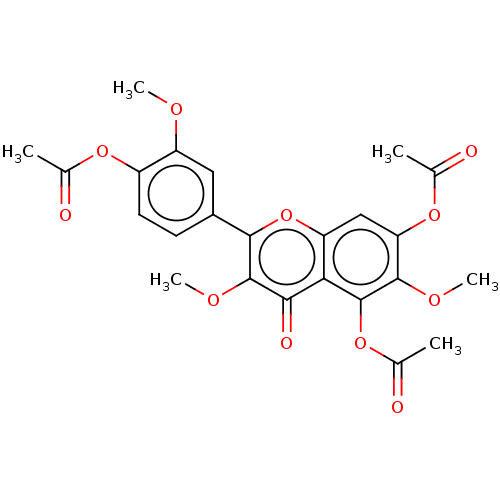 Jaceidin triacetate BDBM645366 US20240016777, Table3.8
Jaceidin triacetate BDBM645366 US20240016777, Table3.8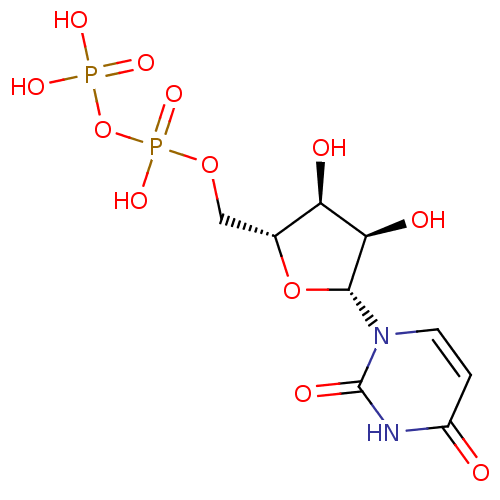 CHEMBL130266 UDP uridine 5'-(trihydrogen diphosphate) BDBM50118239 Uridine diphosphate
CHEMBL130266 UDP uridine 5'-(trihydrogen diphosphate) BDBM50118239 Uridine diphosphate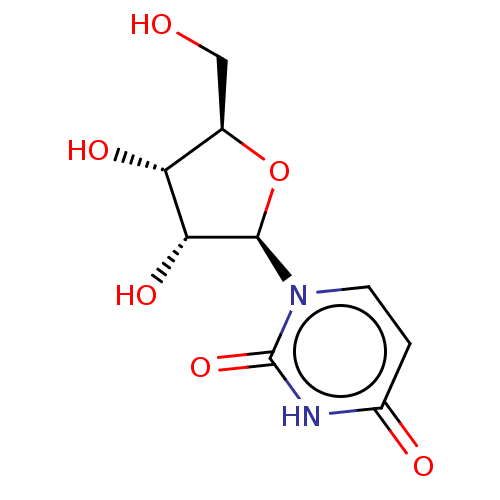 BDBM50088517 Uridine CHEBI:16704
BDBM50088517 Uridine CHEBI:16704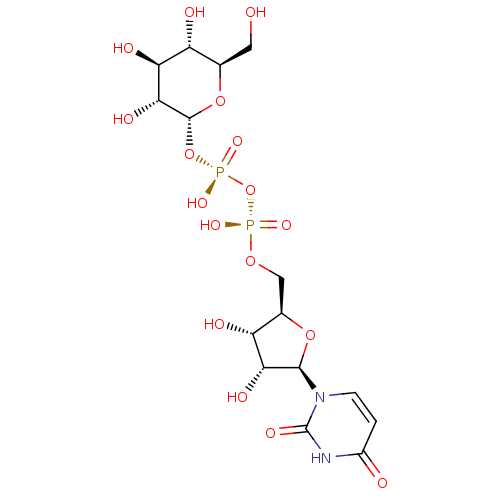 UDPG BDBM50423218 URIDINE DIPHOSPHATE GLUCOSE Udp-Glucose 5''-Diphosphoglucose Uridine-5''-Diphosphoglucose
UDPG BDBM50423218 URIDINE DIPHOSPHATE GLUCOSE Udp-Glucose 5''-Diphosphoglucose Uridine-5''-Diphosphoglucose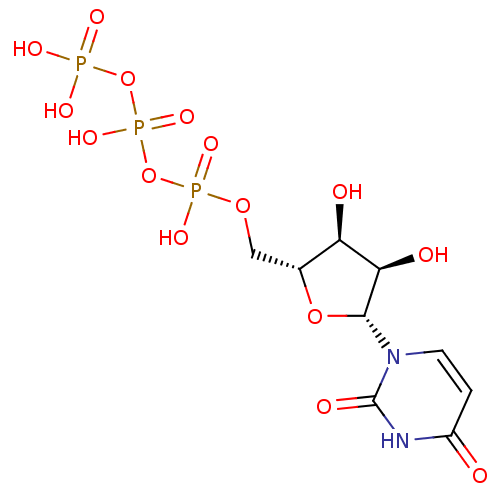 BDBM50118213 UTP uridine 5'-triphosphoric acid H4utp CHEMBL336296 uridine 5'-(tetrahydrogen triphosphate) 5'-UTP
BDBM50118213 UTP uridine 5'-triphosphoric acid H4utp CHEMBL336296 uridine 5'-(tetrahydrogen triphosphate) 5'-UTP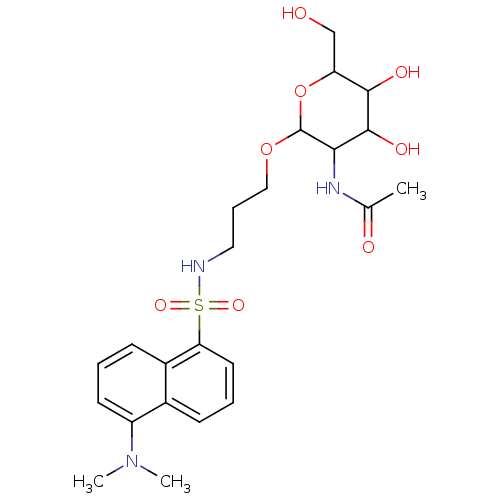 BDBM50173721 Uridine-5'-diphosphogalactose derivative CHEMBL196432
BDBM50173721 Uridine-5'-diphosphogalactose derivative CHEMBL196432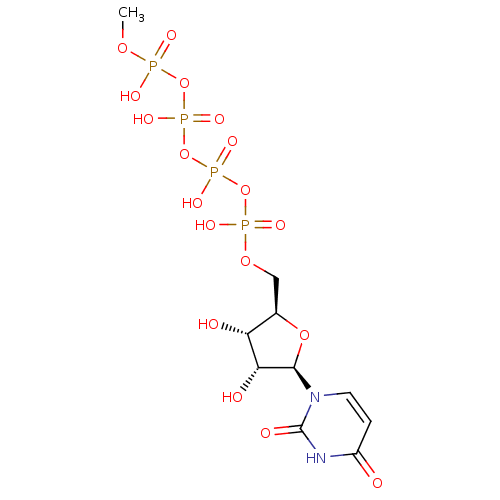 BDBM50271289 Uridine-5'-methyl-tetraphosphate CHEMBL519991
BDBM50271289 Uridine-5'-methyl-tetraphosphate CHEMBL519991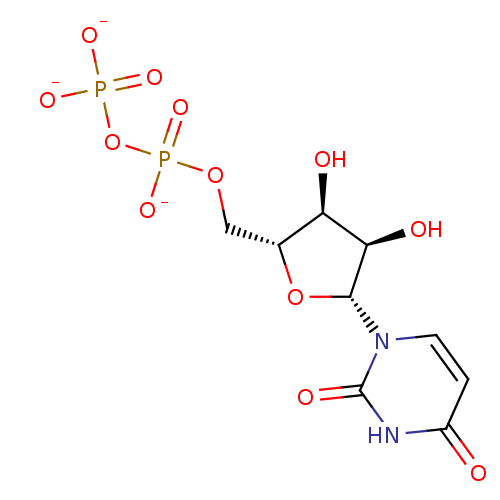 CHEMBL1096401 BDBM50318022 uridine diphosphate trisodium salt
CHEMBL1096401 BDBM50318022 uridine diphosphate trisodium salt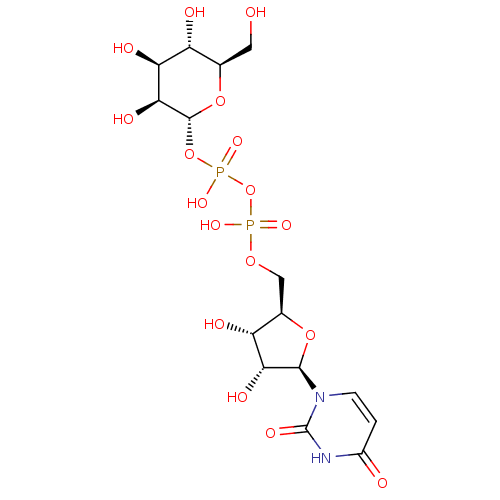 CHEMBL227711 BDBM50209666 URIDINE-5'-DIPHOSPHATE-MANNOSE
CHEMBL227711 BDBM50209666 URIDINE-5'-DIPHOSPHATE-MANNOSE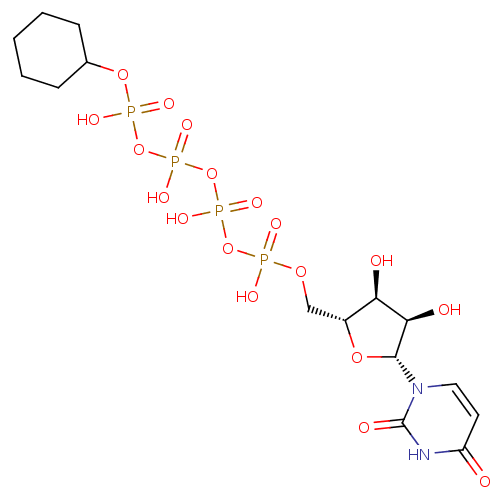 CHEMBL474887 BDBM50270542 Uridine-5'-cyclohexane-tetraphosphate
CHEMBL474887 BDBM50270542 Uridine-5'-cyclohexane-tetraphosphate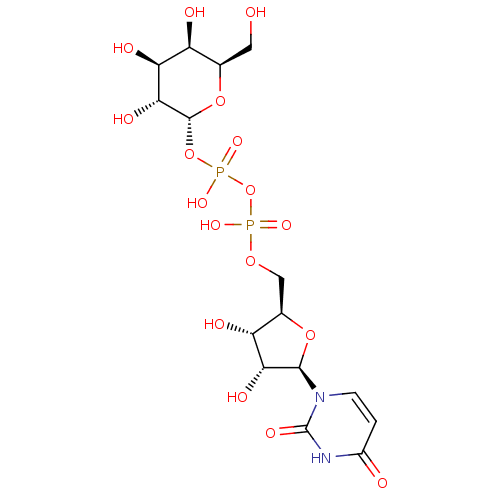 Galactose-Uridine-5'-Diphosphate CHEMBL439009 BDBM50209668
Galactose-Uridine-5'-Diphosphate CHEMBL439009 BDBM50209668 Uridine-5'-phenyl-tetraphosphate BDBM50270541 CHEMBL474886
Uridine-5'-phenyl-tetraphosphate BDBM50270541 CHEMBL474886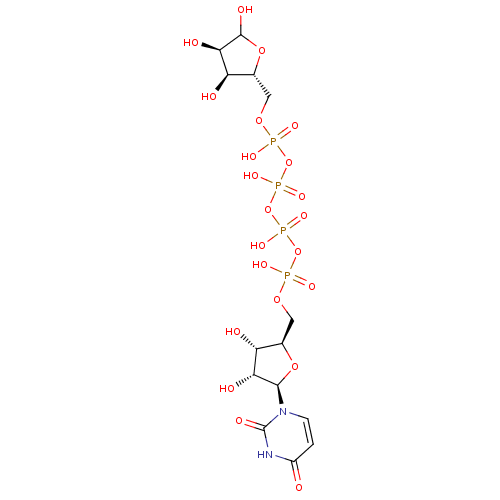 BDBM50205418 CHEMBL410594 uridine 5'-tetraphosphate 5'-ribose
BDBM50205418 CHEMBL410594 uridine 5'-tetraphosphate 5'-ribose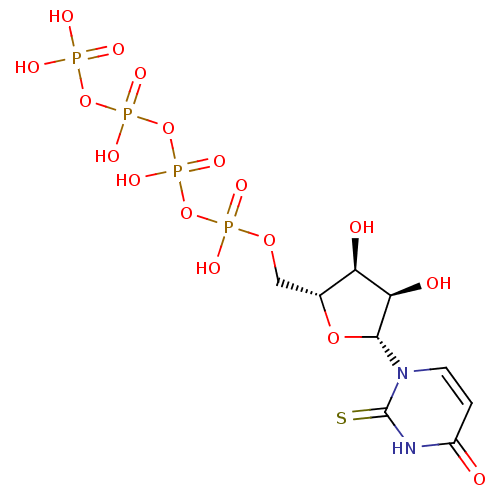 BDBM50271252 2-Thio-uridine-5'-tetraphosphate CHEMBL482485
BDBM50271252 2-Thio-uridine-5'-tetraphosphate CHEMBL482485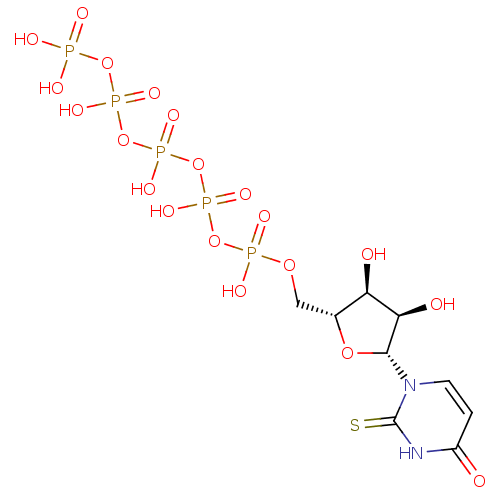 BDBM50271254 CHEMBL482683 2-Thio-uridine-5'-pentaphosphate
BDBM50271254 CHEMBL482683 2-Thio-uridine-5'-pentaphosphate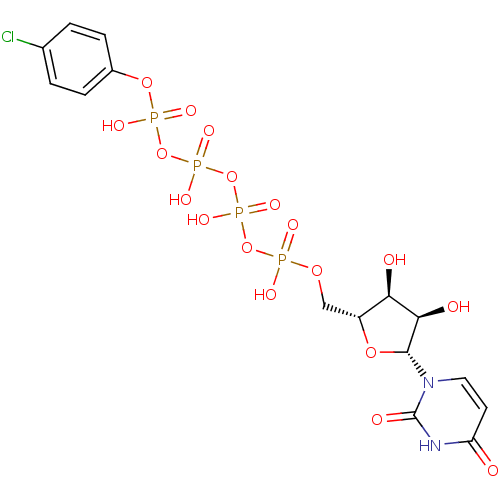 BDBM50345487 Uridine-5'-(4-chlorophenyl)-tetraphosphate CHEMBL1784895
BDBM50345487 Uridine-5'-(4-chlorophenyl)-tetraphosphate CHEMBL1784895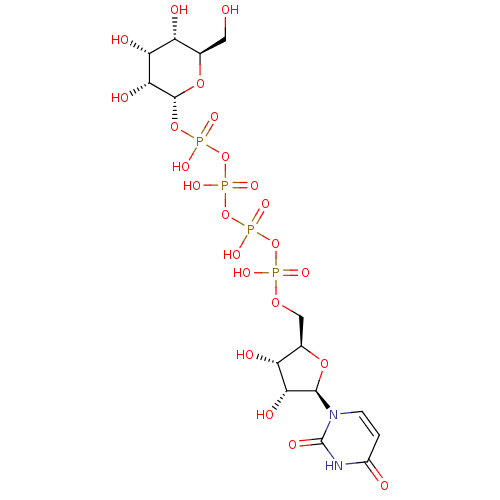 BDBM50345491 Uridine-5'-allose-1'-tetraphosphate CHEMBL1784885
BDBM50345491 Uridine-5'-allose-1'-tetraphosphate CHEMBL1784885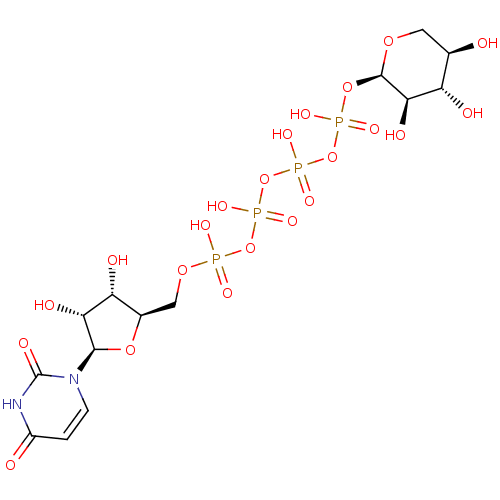 BDBM50345493 CHEMBL1784887 Uridine-5'-xylose-1'-tetraphosphate
BDBM50345493 CHEMBL1784887 Uridine-5'-xylose-1'-tetraphosphate CHEMBL1784900 Uridine-5'-(3-nitrophenyl)-tetraphosphate BDBM50345490
CHEMBL1784900 Uridine-5'-(3-nitrophenyl)-tetraphosphate BDBM50345490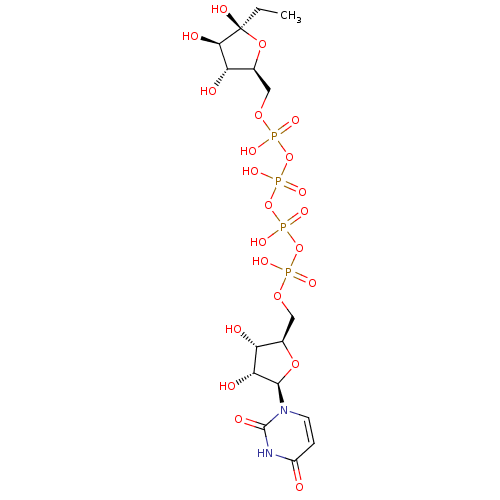 CHEMBL500840 BDBM50270544 Uridine-5'-fructose-6'-tetraphosphate
CHEMBL500840 BDBM50270544 Uridine-5'-fructose-6'-tetraphosphate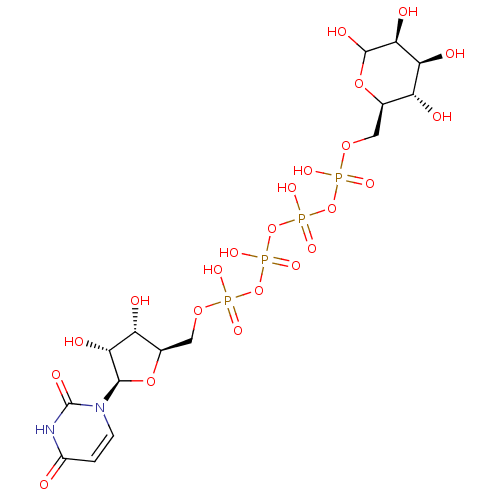 CHEMBL508122 Uridine-5'-mannose-6'-tetraphosphate BDBM50270547
CHEMBL508122 Uridine-5'-mannose-6'-tetraphosphate BDBM50270547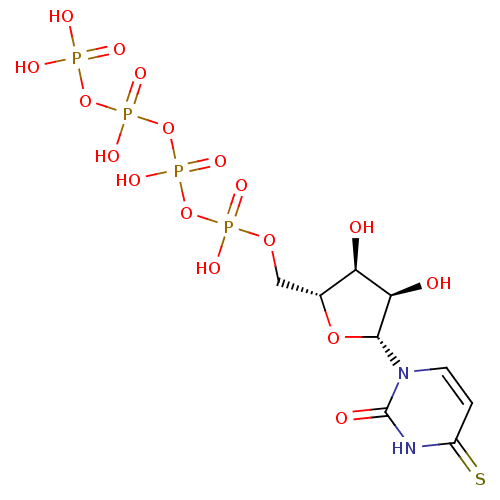 CHEMBL521451 BDBM50271253 4-Thio-uridine-5'-tetraphosphate
CHEMBL521451 BDBM50271253 4-Thio-uridine-5'-tetraphosphate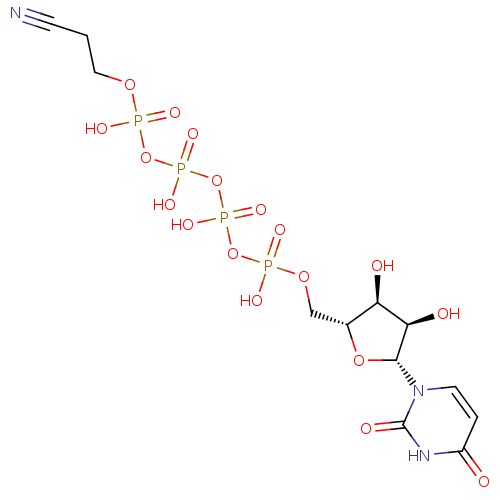 Uridine-5'-(2-cyanoethyl)-tetraphosphate BDBM50271291 CHEMBL484105
Uridine-5'-(2-cyanoethyl)-tetraphosphate BDBM50271291 CHEMBL484105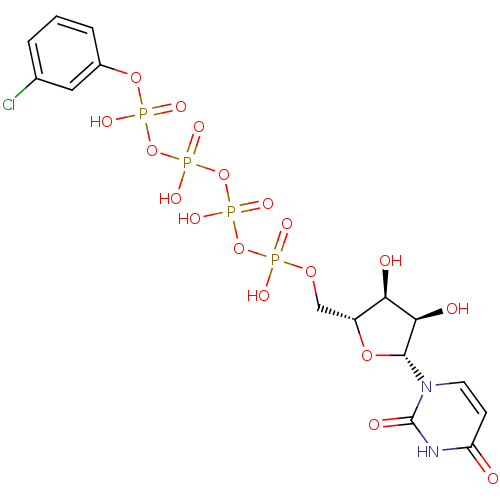 Uridine-5'-(3-chlorophenyl)-tetraphosphate BDBM50345488 CHEMBL1784896
Uridine-5'-(3-chlorophenyl)-tetraphosphate BDBM50345488 CHEMBL1784896 Uridine-5'-(4-nitrophenyl)-tetraphosphate CHEMBL1784899 BDBM50345489
Uridine-5'-(4-nitrophenyl)-tetraphosphate CHEMBL1784899 BDBM50345489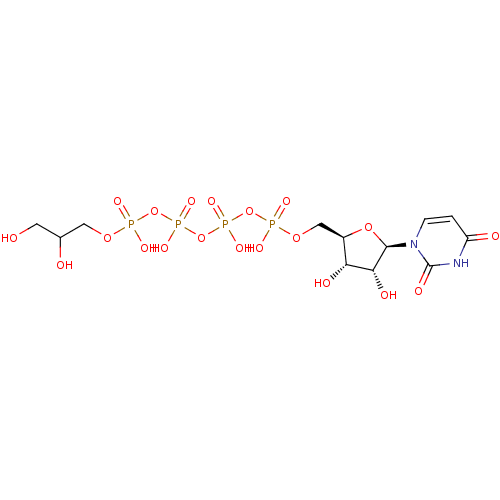 Uridine-5'-alpha-glycerol-tetraphosphate CHEMBL482895 BDBM50271292
Uridine-5'-alpha-glycerol-tetraphosphate CHEMBL482895 BDBM50271292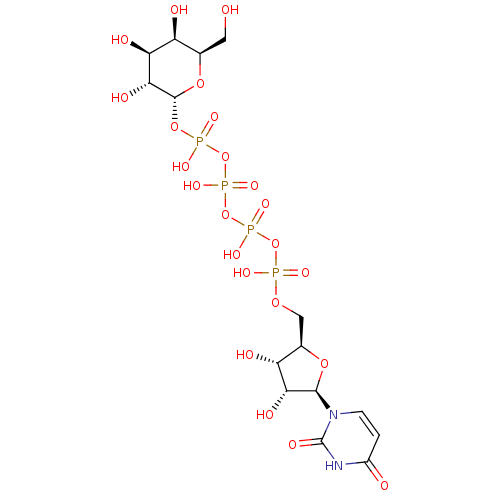 Uridine-5'-galactose-1'-tetraphosphate BDBM50270546 CHEMBL444212
Uridine-5'-galactose-1'-tetraphosphate BDBM50270546 CHEMBL444212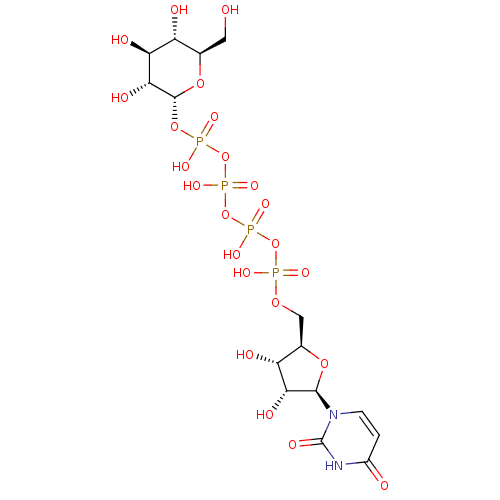 Uridine-5'-glucose-1'-tetraphosphate BDBM50270545 CHEMBL499138
Uridine-5'-glucose-1'-tetraphosphate BDBM50270545 CHEMBL499138 Uridine-5'-mannose-1'-tetraphosphate BDBM50345492 CHEMBL1784886
Uridine-5'-mannose-1'-tetraphosphate BDBM50345492 CHEMBL1784886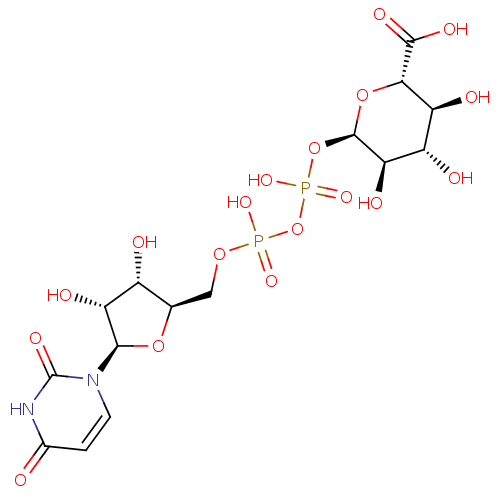 CHEMBL228057 uridine 5'-[3-(alpha-D-glucopyranuronosyl) dihydrogen diphosphate] BDBM50209665 uridine diphosphate glucuronic acid UDP-alpha-D-glucuronic acid
CHEMBL228057 uridine 5'-[3-(alpha-D-glucopyranuronosyl) dihydrogen diphosphate] BDBM50209665 uridine diphosphate glucuronic acid UDP-alpha-D-glucuronic acid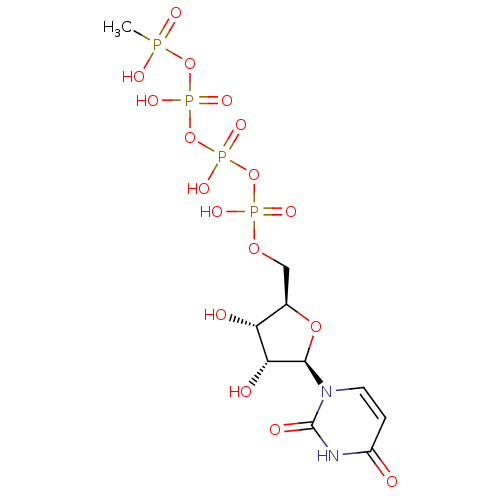 CHEMBL483913 Uridine-5'-methyl(C-P)-tetraphosphate BDBM50271290
CHEMBL483913 Uridine-5'-methyl(C-P)-tetraphosphate BDBM50271290 CHEMBL521487 BDBM50271224 2'-C-Methyl-uridine-5'-triphosphate
CHEMBL521487 BDBM50271224 2'-C-Methyl-uridine-5'-triphosphate P1-Uridine 5'-P3-Cyclohexyltriphosphate TriethylammoniumSalt CHEMBL1083258 BDBM50319138
P1-Uridine 5'-P3-Cyclohexyltriphosphate TriethylammoniumSalt CHEMBL1083258 BDBM50319138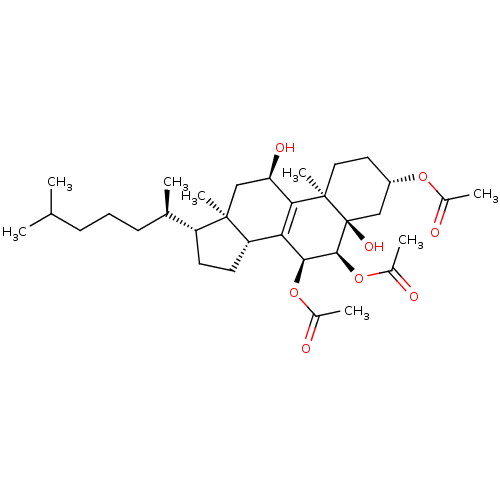 CHEMBL451438 cholest-8-ene-3beta,5alpha,6alpha,7alpha,10alpha-pentol 3,6,7-triacetate BDBM50269197
CHEMBL451438 cholest-8-ene-3beta,5alpha,6alpha,7alpha,10alpha-pentol 3,6,7-triacetate BDBM50269197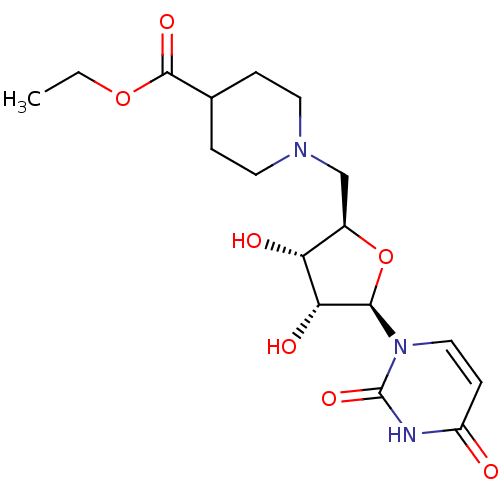 5'-deoxy-5'-N-(ethyl isonipecotatyl)uridine BDBM50292715 CHEMBL445773
5'-deoxy-5'-N-(ethyl isonipecotatyl)uridine BDBM50292715 CHEMBL445773 BDBM50271188 CHEMBL482472 2-Thio-4-methylthio-uridine-5'-triphosphate
BDBM50271188 CHEMBL482472 2-Thio-4-methylthio-uridine-5'-triphosphate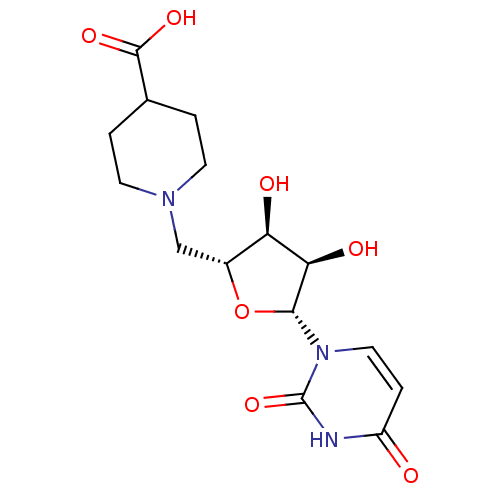 BDBM50342006 5'-Deoxy-5'-N-(4-carboxypiperidinyl)uridine CHEMBL1765476
BDBM50342006 5'-Deoxy-5'-N-(4-carboxypiperidinyl)uridine CHEMBL1765476 CHEMBL1083259 BDBM50319137 P1-Uridine 5'-P3-Phenyltriphosphate Triethylammonium Salt
CHEMBL1083259 BDBM50319137 P1-Uridine 5'-P3-Phenyltriphosphate Triethylammonium Salt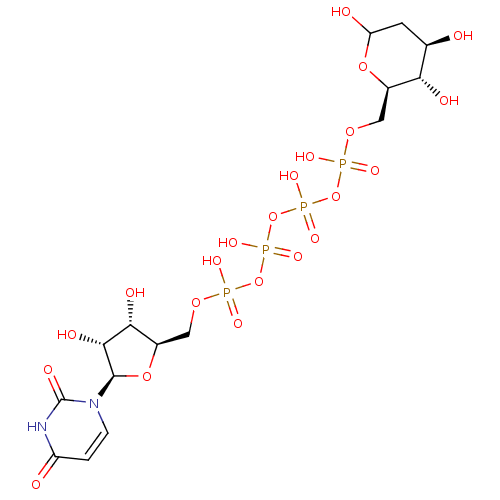 CHEMBL454230 Uridine-5'-(2'-deoxy-glucose)-6'-tetraphosphate BDBM50270548
CHEMBL454230 Uridine-5'-(2'-deoxy-glucose)-6'-tetraphosphate BDBM50270548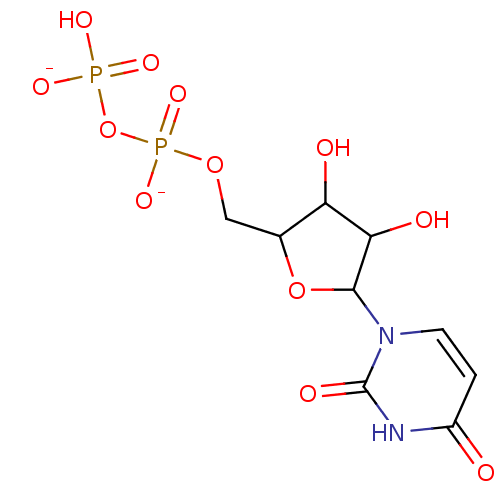 hydrogen ({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl phosphonato}oxy)phosphonate BDBM50096295 Uridine dinucleoside 5'-polyphosphate analogue Uridine
hydrogen ({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl phosphonato}oxy)phosphonate BDBM50096295 Uridine dinucleoside 5'-polyphosphate analogue Uridine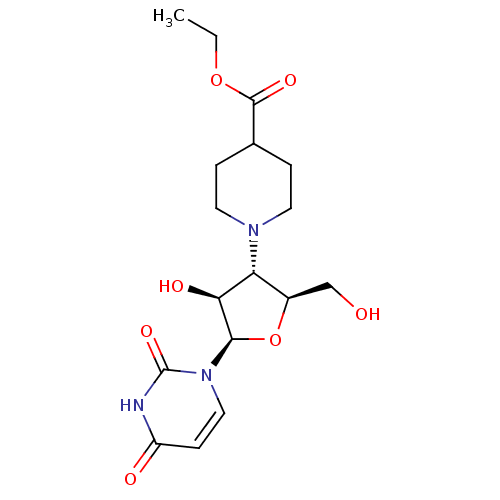 3'-[1-(4-ethoxycarbonylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL471836 BDBM50276040
3'-[1-(4-ethoxycarbonylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL471836 BDBM50276040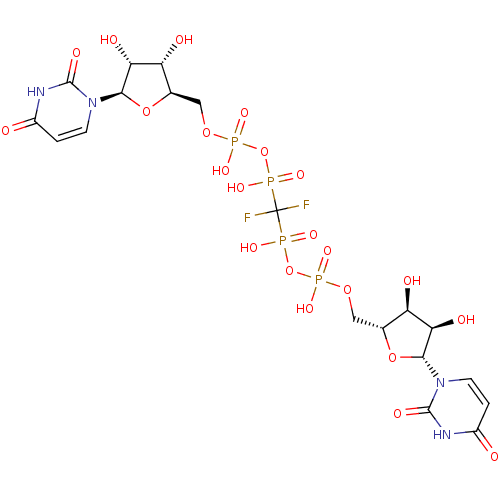 BDBM50270551 P1,P4-di(uridine 5'-)beta,gamma-difluoromethylenetetraphosphate CHEMBL502712
BDBM50270551 P1,P4-di(uridine 5'-)beta,gamma-difluoromethylenetetraphosphate CHEMBL502712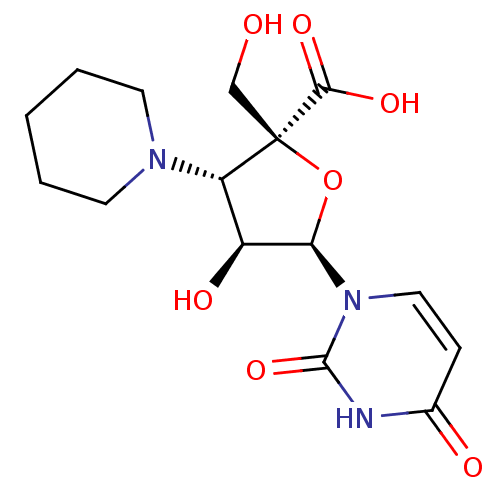 BDBM50292714 CHEMBL461790 3'-N-piperidine-4-carboxyl-3'-deoxyara-uridine
BDBM50292714 CHEMBL461790 3'-N-piperidine-4-carboxyl-3'-deoxyara-uridine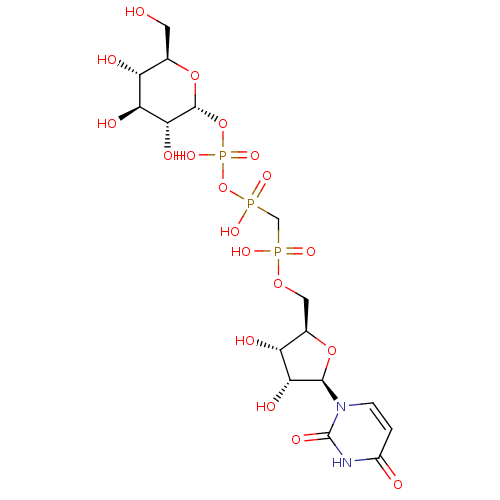 BDBM50319134 Uridine 5'-Glucose-1'-alpha,beta-methylenetriphosphate TriethylammoniumSalt CHEMBL1083263
BDBM50319134 Uridine 5'-Glucose-1'-alpha,beta-methylenetriphosphate TriethylammoniumSalt CHEMBL1083263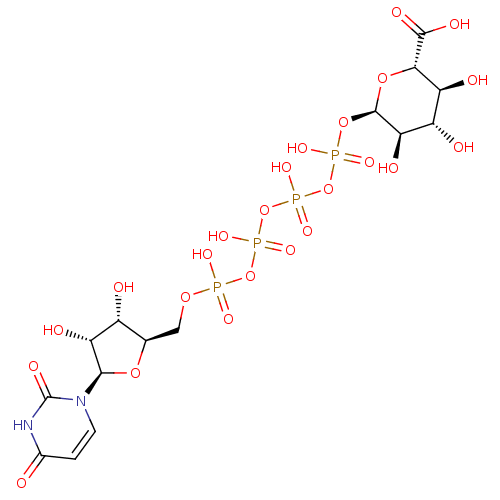 CHEMBL1784902 BDBM50345495 P1-(Uridine-5'-)P4-(glucuronic acid-1'-)tetraphosphate
CHEMBL1784902 BDBM50345495 P1-(Uridine-5'-)P4-(glucuronic acid-1'-)tetraphosphate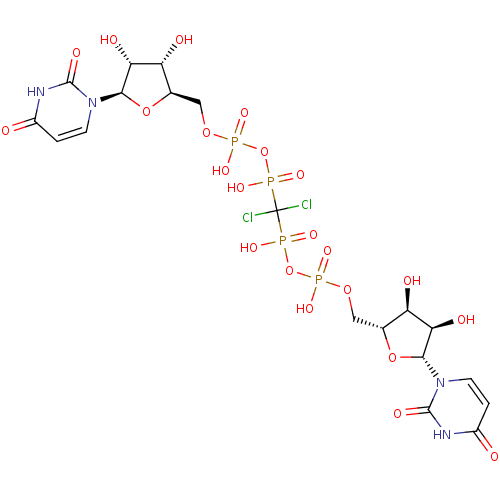 P1,P4-di(uridine 5'-)beta,gamma-dichloromethlyenetetraphosphate BDBM50270552 CHEMBL476021
P1,P4-di(uridine 5'-)beta,gamma-dichloromethlyenetetraphosphate BDBM50270552 CHEMBL476021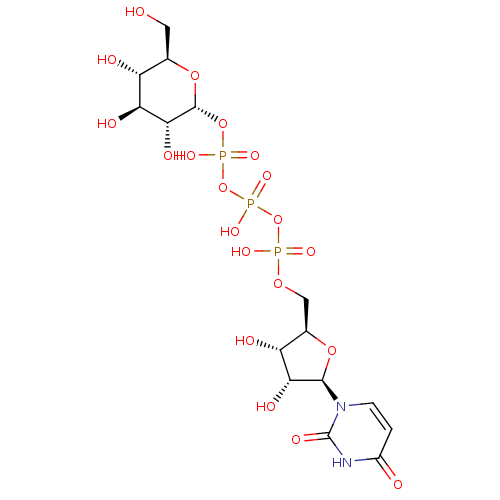 P1-Uridine 5'-P3-[1]Glucose-1'-Triphosphate TriethylammoniumSalt BDBM50319132 CHEMBL1083260
P1-Uridine 5'-P3-[1]Glucose-1'-Triphosphate TriethylammoniumSalt BDBM50319132 CHEMBL1083260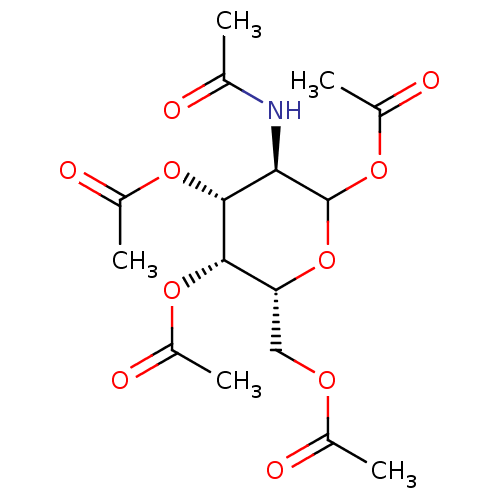 (3R,4R,5R,6R)-3-acetamido-6-(acetoxymethyl)tetrahydro-2H-pyran-2,4,5-triyl triacetate CHEMBL610736 BDBM50304486
(3R,4R,5R,6R)-3-acetamido-6-(acetoxymethyl)tetrahydro-2H-pyran-2,4,5-triyl triacetate CHEMBL610736 BDBM50304486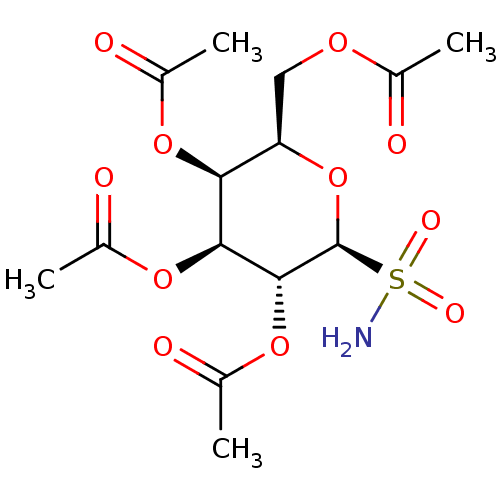 BDBM50299574 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL579226
BDBM50299574 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL579226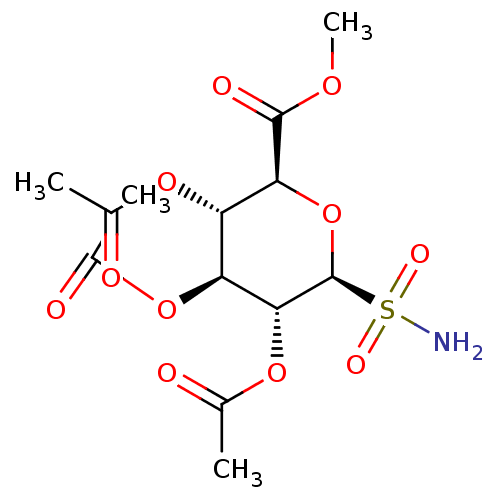 BDBM50299575 (2S,3S,4S,5R,6S)-2-(methoxycarbonyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL584312
BDBM50299575 (2S,3S,4S,5R,6S)-2-(methoxycarbonyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL584312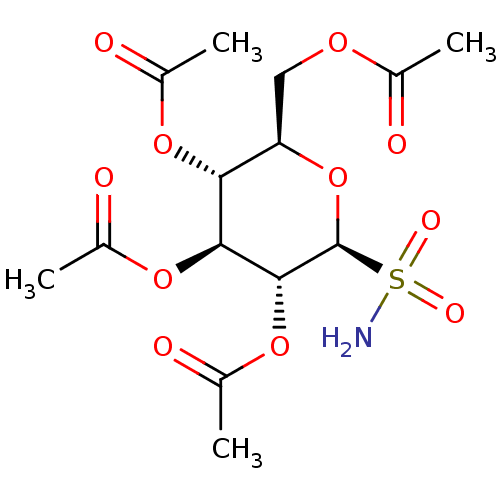 CHEMBL568705 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50299573
CHEMBL568705 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50299573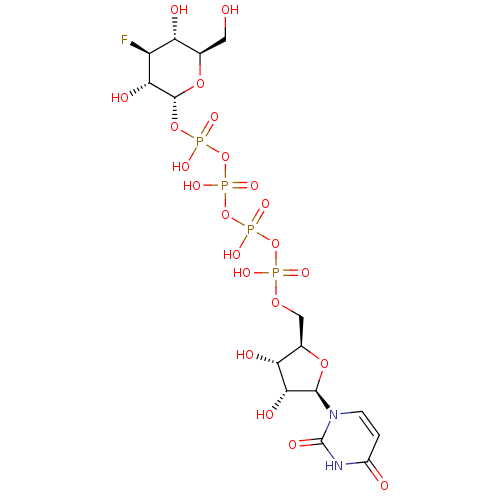 BDBM50345496 P1-(Uridine-5'-)P4-(3'-deoxy-3'-fluoroglucose-1'-)tetraphosphate CHEMBL1784904
BDBM50345496 P1-(Uridine-5'-)P4-(3'-deoxy-3'-fluoroglucose-1'-)tetraphosphate CHEMBL1784904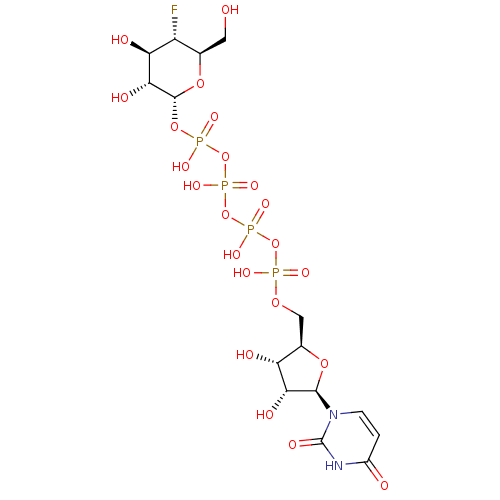 CHEMBL1784293 P1-(Uridine-5'-)P4-(4'-deoxy-4'-fluoroglucose-1'-)tetraphosphate BDBM50345499
CHEMBL1784293 P1-(Uridine-5'-)P4-(4'-deoxy-4'-fluoroglucose-1'-)tetraphosphate BDBM50345499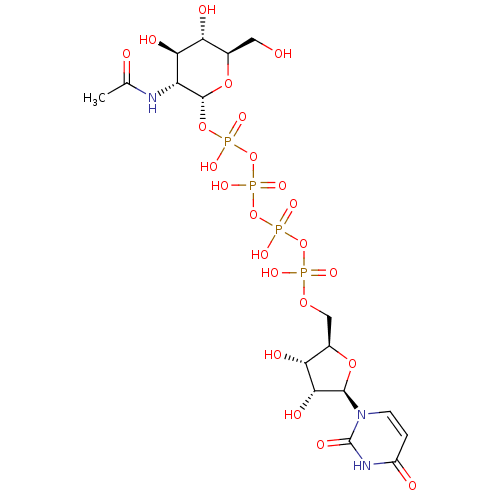 P1-(Uridine-5'-)P4-(2'-deoxy-2'-acetamidoglucose-1'-)tetraphosphate CHEMBL1784889 BDBM50345494
P1-(Uridine-5'-)P4-(2'-deoxy-2'-acetamidoglucose-1'-)tetraphosphate CHEMBL1784889 BDBM50345494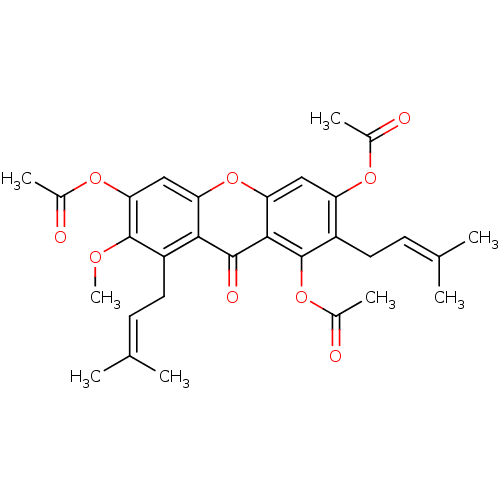 CHEMBL1782243 7-methoxy-2,8-bis(3-methylbut-2-enyl)-9-oxo-9H-xanthene-1,3,6-triyl triacetate BDBM50346338
CHEMBL1782243 7-methoxy-2,8-bis(3-methylbut-2-enyl)-9-oxo-9H-xanthene-1,3,6-triyl triacetate BDBM50346338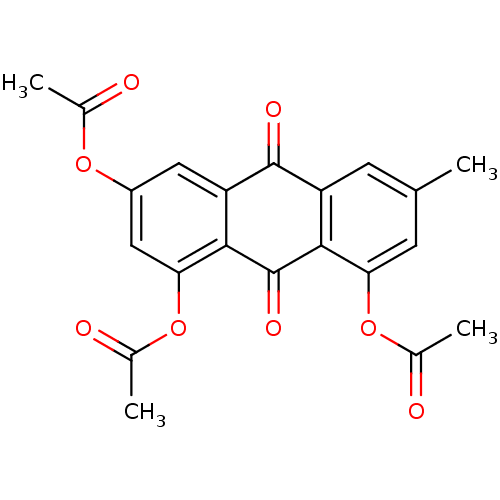 CHEMBL44247 Acetic acid 3,8-diacetoxy-6-methyl-9,10-dioxo-9,10-dihydro-anthracen-1-yl ester emodin triacetate BDBM50005914
CHEMBL44247 Acetic acid 3,8-diacetoxy-6-methyl-9,10-dioxo-9,10-dihydro-anthracen-1-yl ester emodin triacetate BDBM50005914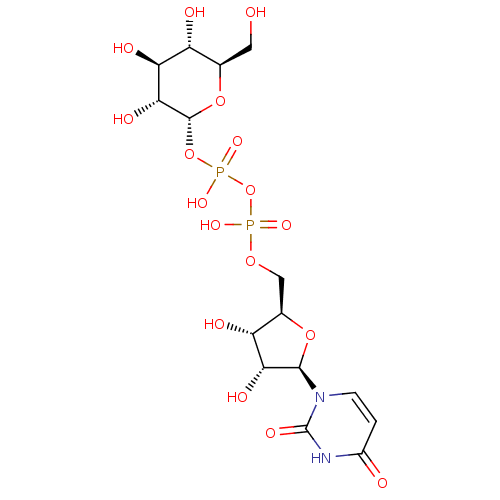 uridine 5'-[3-alpha-D-glucopyranosyl dihydrogen diphosphate] UDP-alpha-D-glucose BDBM50209659
uridine 5'-[3-alpha-D-glucopyranosyl dihydrogen diphosphate] UDP-alpha-D-glucose BDBM50209659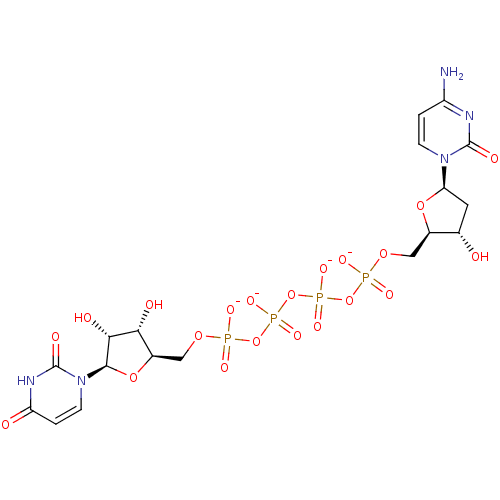 BDBM50341904 CHEMBL1767407 [P(1)-(uridine 5')-P(4)-(2'-deoxycytidine 5')tetraphosphate,tetrasodium salt]
BDBM50341904 CHEMBL1767407 [P(1)-(uridine 5')-P(4)-(2'-deoxycytidine 5')tetraphosphate,tetrasodium salt]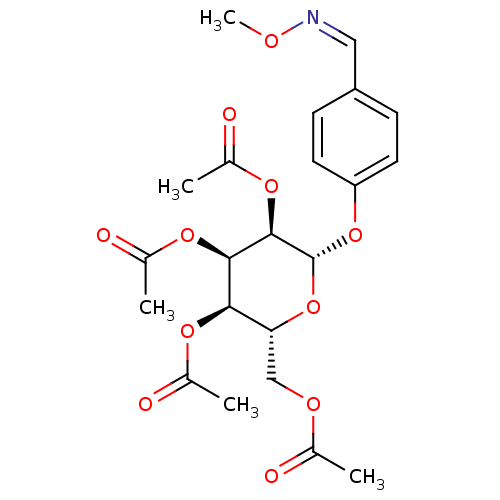 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302735 CHEMBL583541
(2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302735 CHEMBL583541 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302741 CHEMBL569784
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302741 CHEMBL569784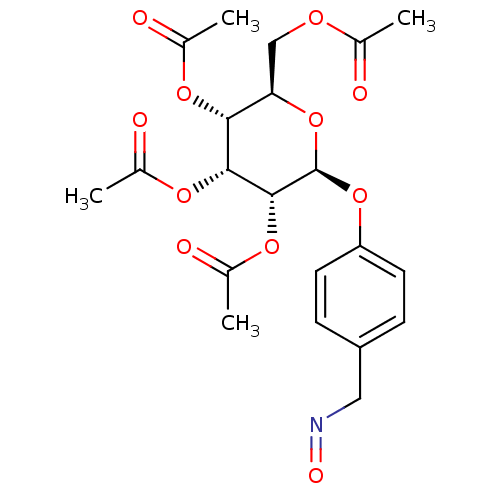 BDBM50302742 CHEMBL569314 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
BDBM50302742 CHEMBL569314 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate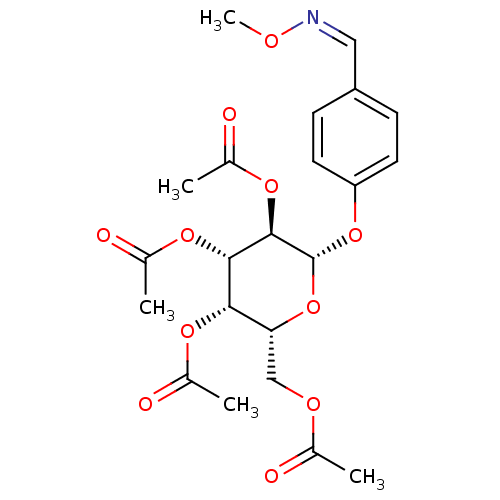 CHEMBL570195 BDBM50302734 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
CHEMBL570195 BDBM50302734 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate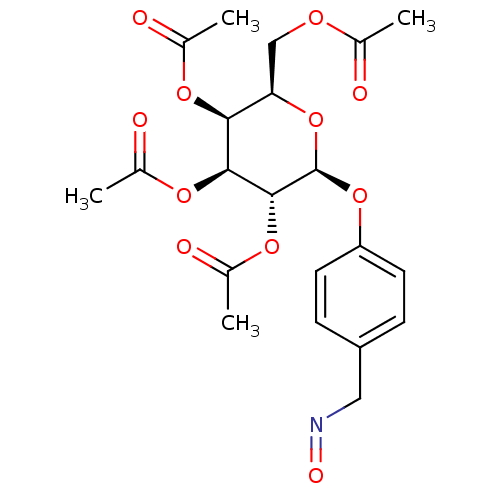 CHEMBL571325 BDBM50302740 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
CHEMBL571325 BDBM50302740 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate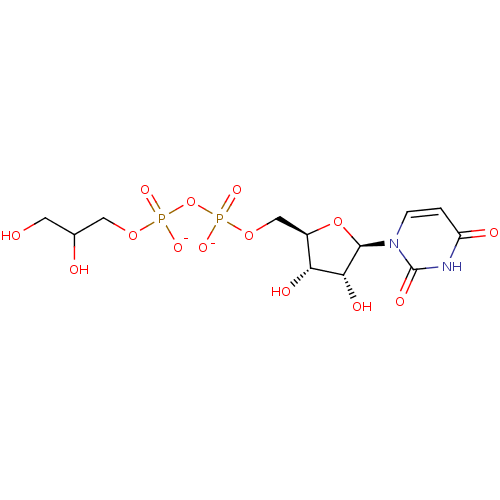 BDBM50303334 Diphosphoric Acid 1-r-Glycerol Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303334 Diphosphoric Acid 1-r-Glycerol Ester 2-(Uridine-5'-yl)ester,di ammonium Salt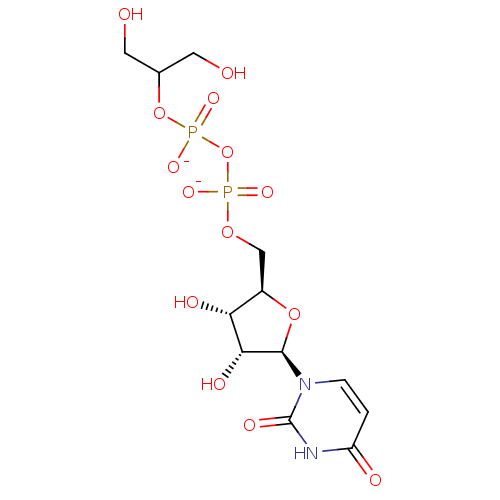 BDBM50303335 Diphosphoric Acid 1-beta-Glycerol Ester 2-(Uridine-5'-yl)ester,bis-Triethylammonium Salt
BDBM50303335 Diphosphoric Acid 1-beta-Glycerol Ester 2-(Uridine-5'-yl)ester,bis-Triethylammonium Salt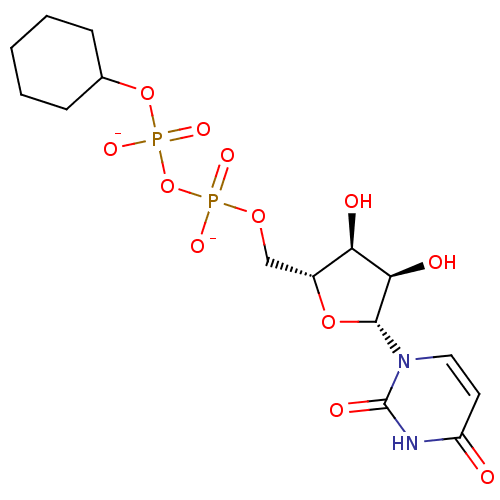 BDBM50303336 Diphosphoric Acid 1-beta-Cyclohexyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303336 Diphosphoric Acid 1-beta-Cyclohexyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt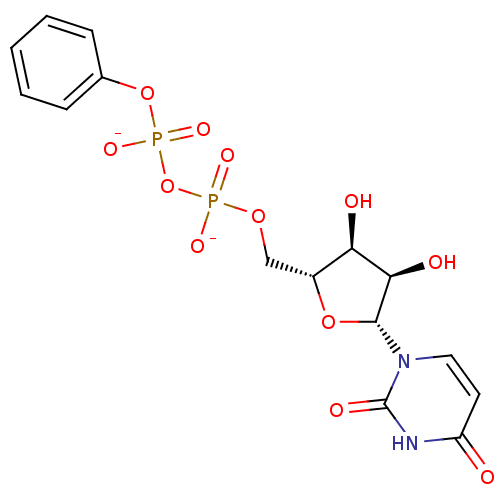 BDBM50303348 Diphosphoric Acid 1-beta-Phenyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303348 Diphosphoric Acid 1-beta-Phenyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt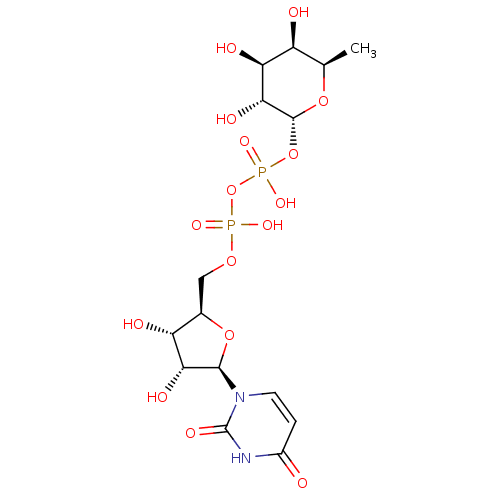 BDBM50304031 Diphosphoric Acid 1''-alpha-D-[1'']Fucopyranosyl Ester 2-(uridine-5'-yl)ester CHEMBL595940
BDBM50304031 Diphosphoric Acid 1''-alpha-D-[1'']Fucopyranosyl Ester 2-(uridine-5'-yl)ester CHEMBL595940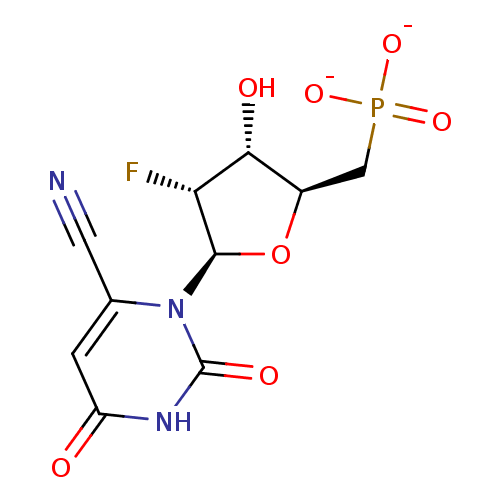 BDBM50341905 CHEMBL1765124 ammonium 6-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate
BDBM50341905 CHEMBL1765124 ammonium 6-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate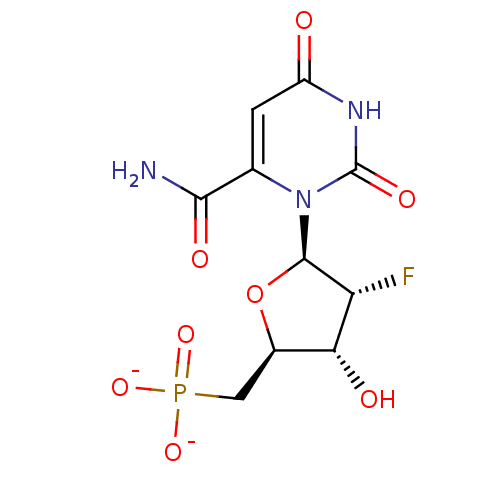 BDBM50341906 Ammonium 6-Amido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate CHEMBL1765121
BDBM50341906 Ammonium 6-Amido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate CHEMBL1765121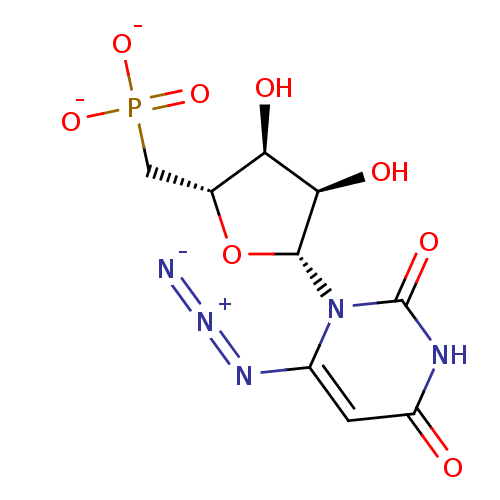 CHEMBL1765126 ammonium 6-Azido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341909
CHEMBL1765126 ammonium 6-Azido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341909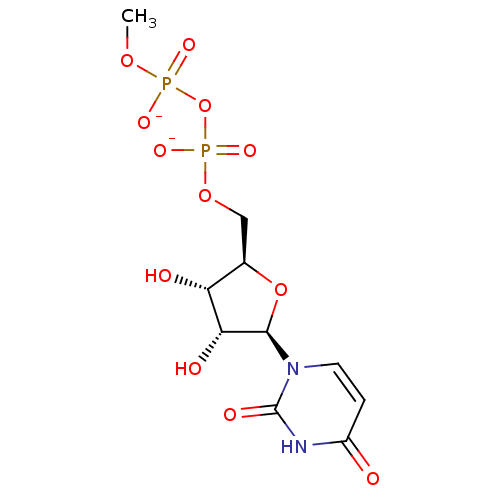 Diphosphoric Acid 1-beta-Methyl Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303341
Diphosphoric Acid 1-beta-Methyl Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303341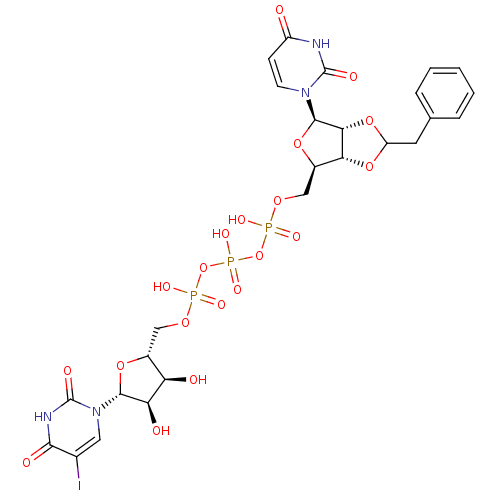 P1-((2-benzyl-1,3-dioxolo-4-yl)uridine 5')P3-(5-iodouridine5') triphosphate CHEMBL503256 BDBM50270549
P1-((2-benzyl-1,3-dioxolo-4-yl)uridine 5')P3-(5-iodouridine5') triphosphate CHEMBL503256 BDBM50270549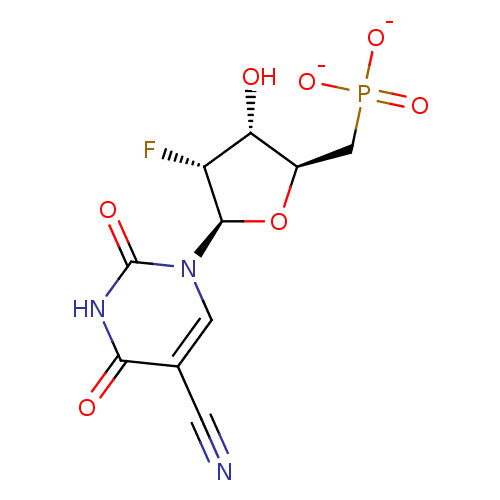 ammonium 5-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341907 CHEMBL1765122
ammonium 5-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341907 CHEMBL1765122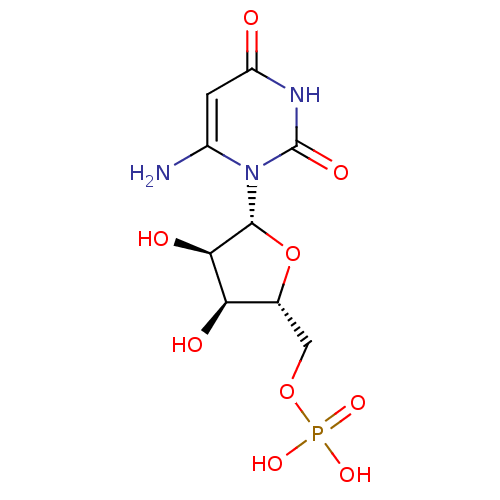 BDBM21338 6-Amino-uridine 5-O-Monophosphate C6-Uridine Derivative, 15 JMC521648 Compound 37 {[(2R,3S,4R,5R)-5-(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
BDBM21338 6-Amino-uridine 5-O-Monophosphate C6-Uridine Derivative, 15 JMC521648 Compound 37 {[(2R,3S,4R,5R)-5-(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid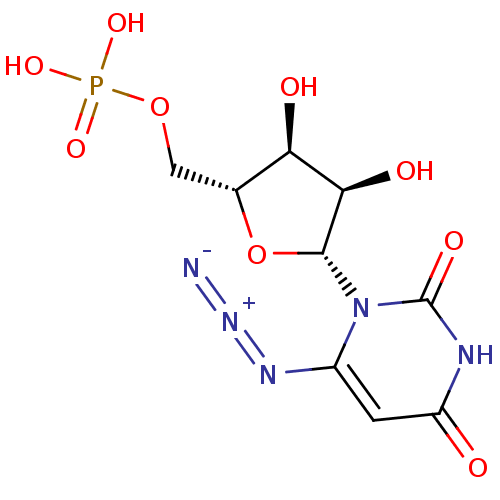 JMC521648 Compound 36 C6-Uridine Derivative, 14 6-Azido-uridine 5-O-Monophosphate BDBM21337 {[(2R,3S,4R,5R)-5-(6-azido-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
JMC521648 Compound 36 C6-Uridine Derivative, 14 6-Azido-uridine 5-O-Monophosphate BDBM21337 {[(2R,3S,4R,5R)-5-(6-azido-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302749 CHEMBL569974
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302749 CHEMBL569974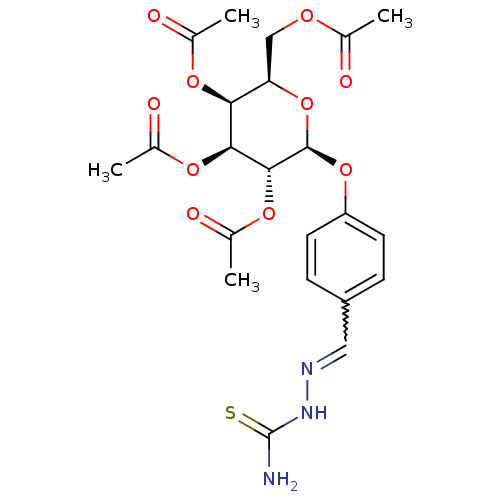 BDBM50302748 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL569079
BDBM50302748 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL569079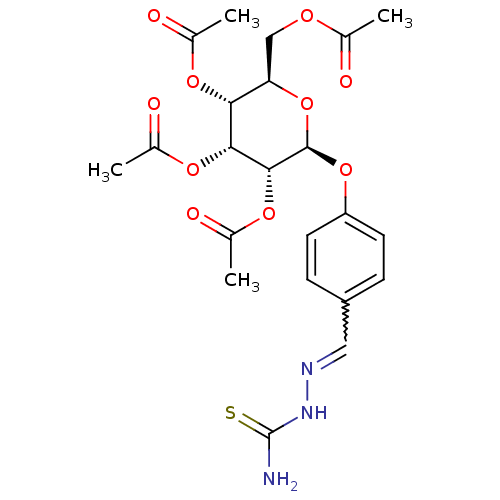 BDBM50302750 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL568781
BDBM50302750 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL568781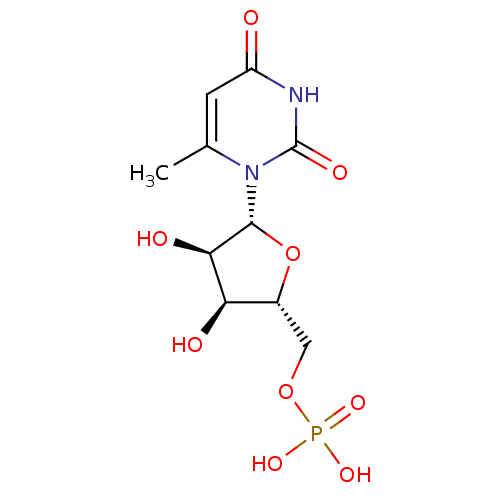 BDBM21339 C6-Uridine Derivative, 16 {[(2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)oxolan-2-yl]methoxy}phosphonic acid JMC521648 Compound 38 6-Methyl-uridine 5-O-Monophosphate
BDBM21339 C6-Uridine Derivative, 16 {[(2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)oxolan-2-yl]methoxy}phosphonic acid JMC521648 Compound 38 6-Methyl-uridine 5-O-Monophosphate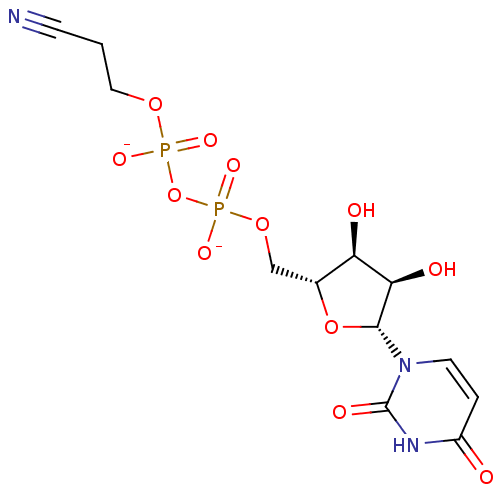 BDBM50303332 Diphosphoric Acid 1-beta-(2-Cyanoethyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt
BDBM50303332 Diphosphoric Acid 1-beta-(2-Cyanoethyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt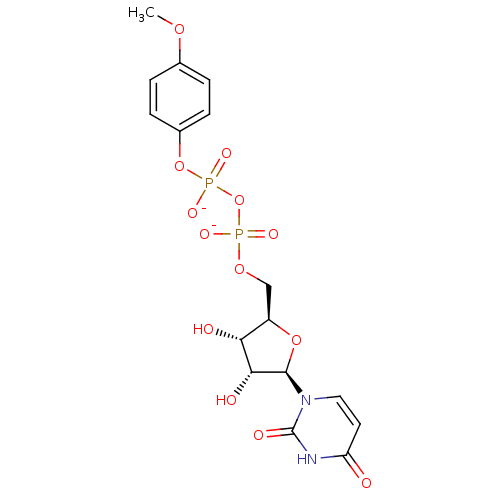 BDBM50303337 Diphosphoric Acid 1-beta-(4-Methoxyphenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303337 Diphosphoric Acid 1-beta-(4-Methoxyphenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt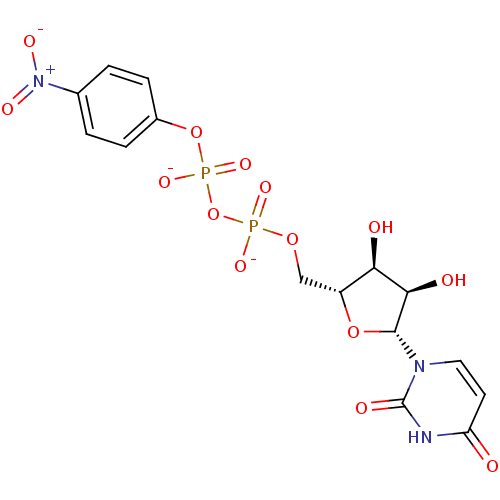 BDBM50303349 Diphosphoric Acid 1-beta-(4-Nitrophenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303349 Diphosphoric Acid 1-beta-(4-Nitrophenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt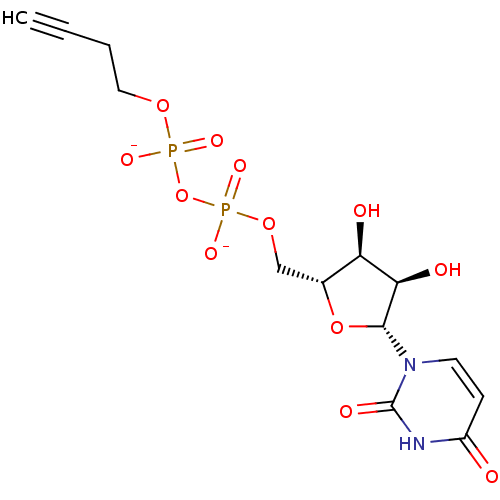 Diphosphoric Acid 1-beta-(3-Butynyl)Ester 2-(Uridine-5'-yl)-ester, bis-Triethylammonium Salt BDBM50303345
Diphosphoric Acid 1-beta-(3-Butynyl)Ester 2-(Uridine-5'-yl)-ester, bis-Triethylammonium Salt BDBM50303345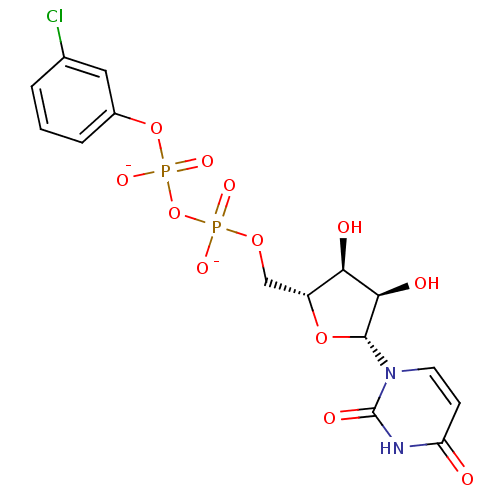 Diphosphoric Acid 1-beta-(3-Chlorophenyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303350
Diphosphoric Acid 1-beta-(3-Chlorophenyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303350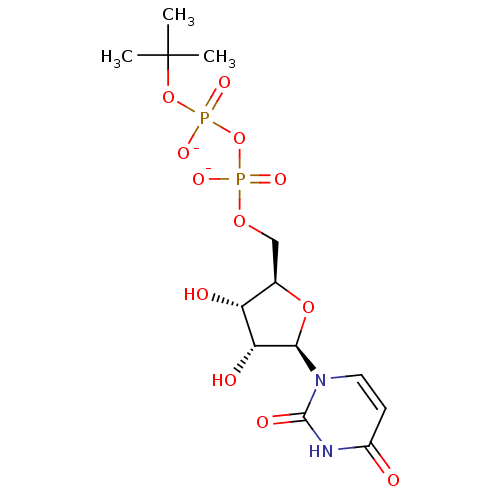 Diphosphoric Acid 1-beta-tert-Butyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303346
Diphosphoric Acid 1-beta-tert-Butyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303346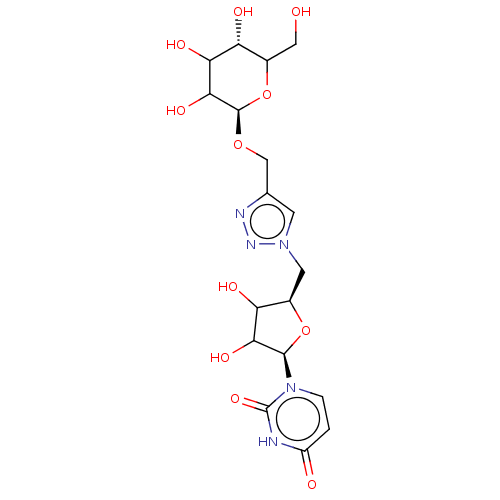 BDBM222427 5'-deoxy-5'-[4-(β-D-glucopyranosyloxymethyl)-1,2-3-triazol-1-yl]uridine (5)
BDBM222427 5'-deoxy-5'-[4-(β-D-glucopyranosyloxymethyl)-1,2-3-triazol-1-yl]uridine (5)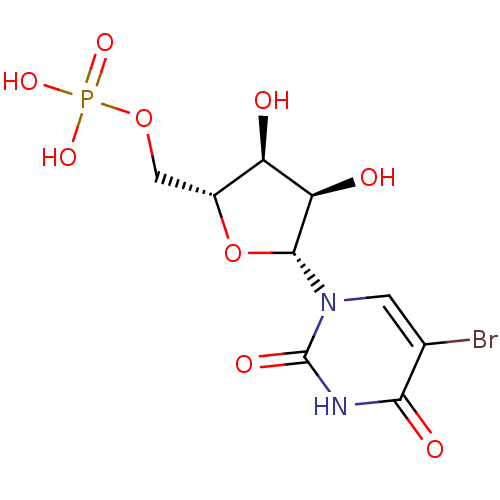 BDBM50199184 CHEMBL214830 5-BROMO-URIDINE-5'-MONOPHOSPHATE 5-bromo-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-dione 5'-monophosphate
BDBM50199184 CHEMBL214830 5-BROMO-URIDINE-5'-MONOPHOSPHATE 5-bromo-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-dione 5'-monophosphate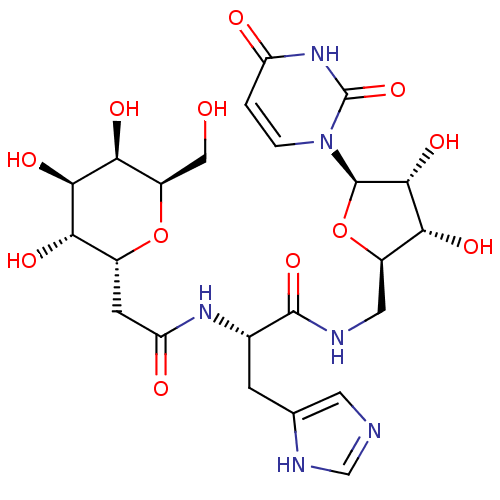 BDBM50333187 CHEMBL1644891 5'-N-[N-alpha-(2,6-Anhydro-D-glycero-L-gluco-heptit-1-yl-acetamido)-histidinamido]-5'-deoxy-uridine
BDBM50333187 CHEMBL1644891 5'-N-[N-alpha-(2,6-Anhydro-D-glycero-L-gluco-heptit-1-yl-acetamido)-histidinamido]-5'-deoxy-uridine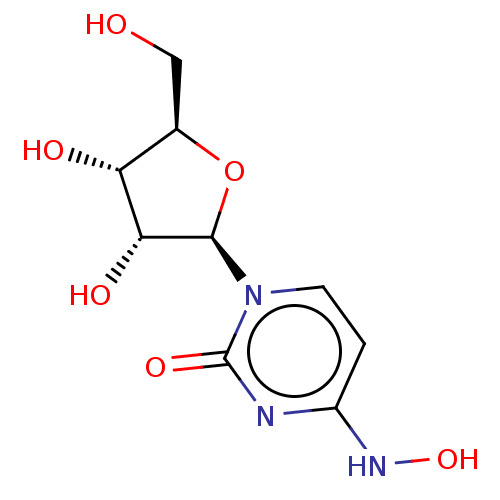 N(4)-Hydroxycytidine Uridine, 4-oxime EIDD-1931 US20240034730, Compound EIDD-1931 Beta-D-N4-hydroxycytidine NHC N4-Hydroxycytidine BDBM430624
N(4)-Hydroxycytidine Uridine, 4-oxime EIDD-1931 US20240034730, Compound EIDD-1931 Beta-D-N4-hydroxycytidine NHC N4-Hydroxycytidine BDBM430624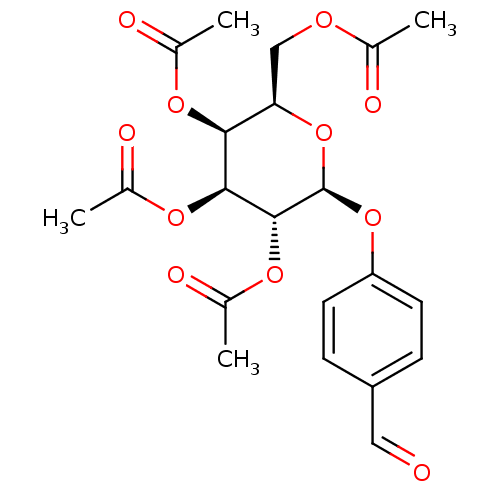 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-galactopyranoside (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251160 CHEMBL465710
4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-galactopyranoside (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251160 CHEMBL465710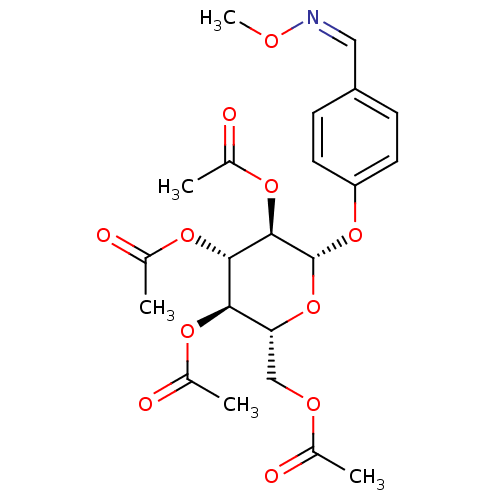 1-(4-(2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl) benzylidene)methoxyamine (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251170 CHEMBL463856
1-(4-(2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl) benzylidene)methoxyamine (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251170 CHEMBL463856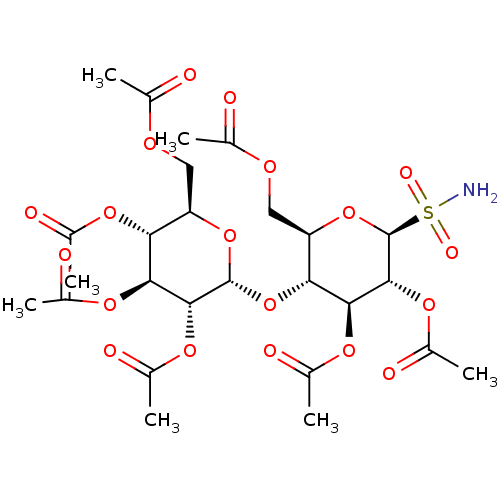 BDBM50299577 (2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573460
BDBM50299577 (2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573460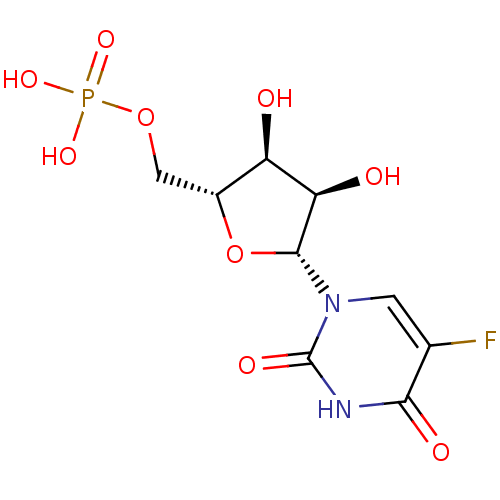 {[(2R,3S,4R,5R)-5-(5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27943 uridine derivative, 39
{[(2R,3S,4R,5R)-5-(5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27943 uridine derivative, 39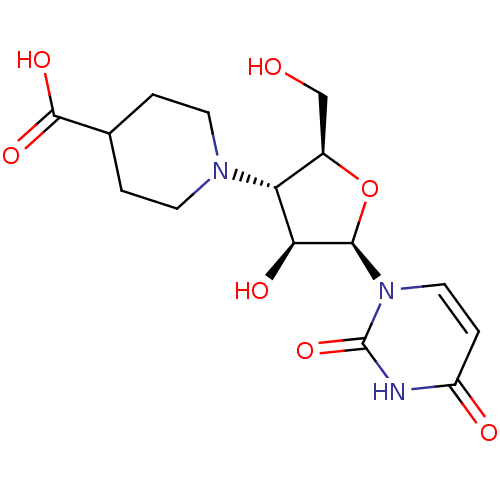 BDBM50276039 3'-[1-(4-carboxylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL469949 1-[3-(4-CARBOXYPIPERIDIN-1-YL)-3-DEOXY-BETA-D-ARABINOFURANOSYL]PYRIMIDINE-2,4(1H,3H)-DIONE
BDBM50276039 3'-[1-(4-carboxylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL469949 1-[3-(4-CARBOXYPIPERIDIN-1-YL)-3-DEOXY-BETA-D-ARABINOFURANOSYL]PYRIMIDINE-2,4(1H,3H)-DIONE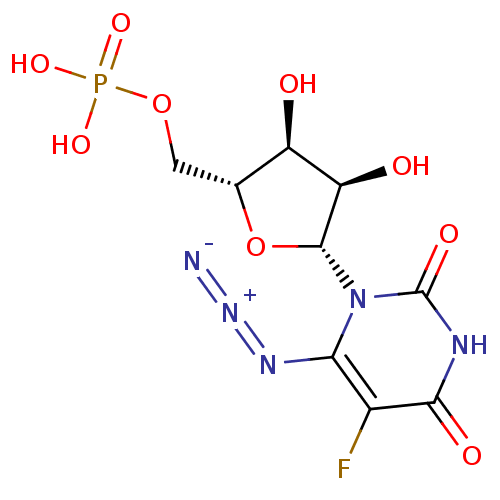 uridine derivative, 41 BDBM27944 {[(2R,3S,4R,5R)-5-(6-azido-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
uridine derivative, 41 BDBM27944 {[(2R,3S,4R,5R)-5-(6-azido-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid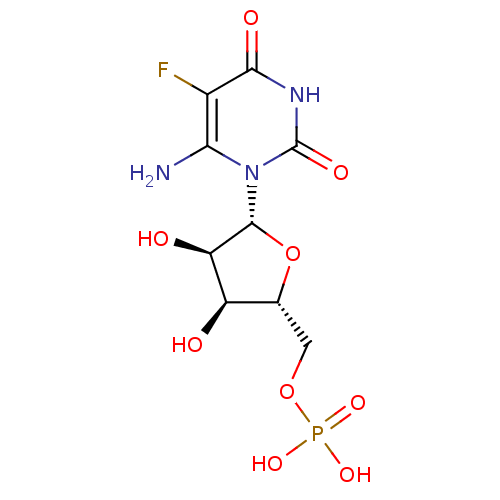 uridine derivative, 42 {[(2R,3S,4R,5R)-5-(6-amino-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27945
uridine derivative, 42 {[(2R,3S,4R,5R)-5-(6-amino-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27945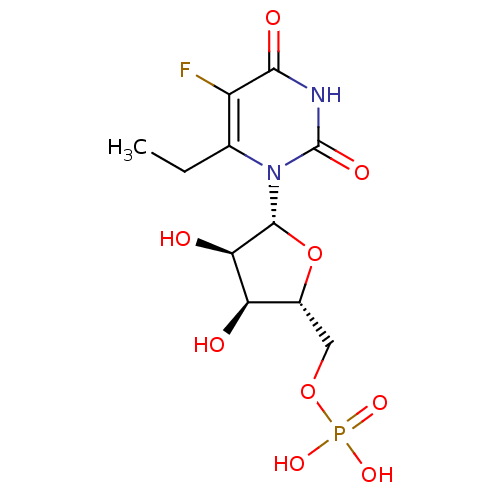 uridine derivative, 43 BDBM27946 {[(2R,3S,4R,5R)-5-(6-ethyl-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
uridine derivative, 43 BDBM27946 {[(2R,3S,4R,5R)-5-(6-ethyl-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid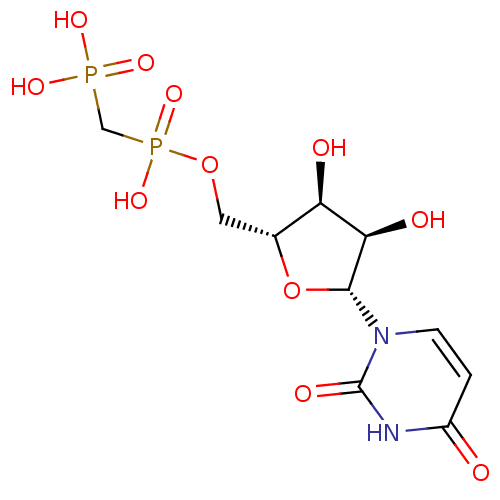 CHEMBL507060 ((((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(hydroxy)phosphoryl)methylphosphonic acid BDBM50271102 Uridine-5'-alpha,beta-methylene-diphosphate {[(2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-3,4-dihydroxy-tetrahydro-furan-2-ylmethoxy]-hydroxy-phosphorylmethyl}-phosphonic acid 5'-O-[(S)-HYDROXY(PHOSPHONOMETHYL)PHOSPHORYL]URIDINE
CHEMBL507060 ((((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(hydroxy)phosphoryl)methylphosphonic acid BDBM50271102 Uridine-5'-alpha,beta-methylene-diphosphate {[(2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-3,4-dihydroxy-tetrahydro-furan-2-ylmethoxy]-hydroxy-phosphorylmethyl}-phosphonic acid 5'-O-[(S)-HYDROXY(PHOSPHONOMETHYL)PHOSPHORYL]URIDINE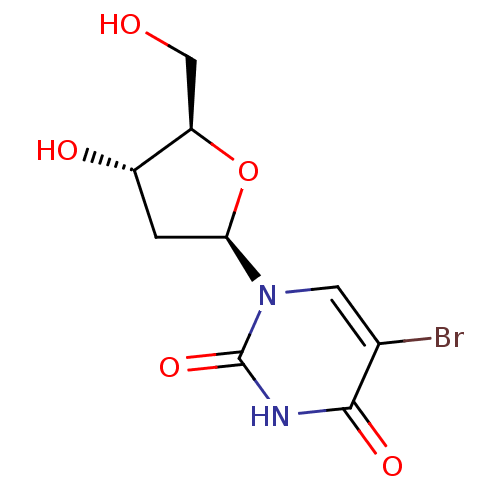 5-bromo-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-BROMO-2'-DEOXY URIDINE cid_6035 Bromodeoxyuridine CHEMBL222280 BDBM50207303
5-bromo-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-BROMO-2'-DEOXY URIDINE cid_6035 Bromodeoxyuridine CHEMBL222280 BDBM50207303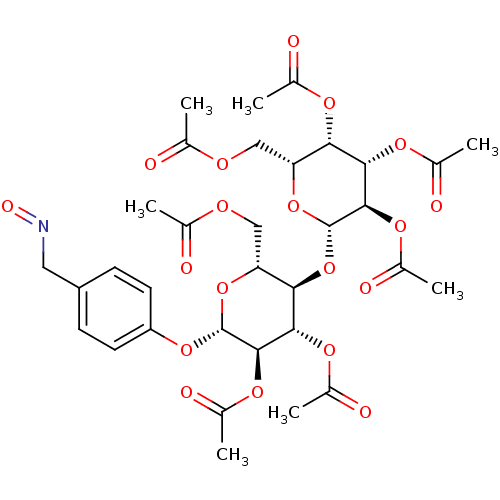 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302737 CHEMBL577106
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302737 CHEMBL577106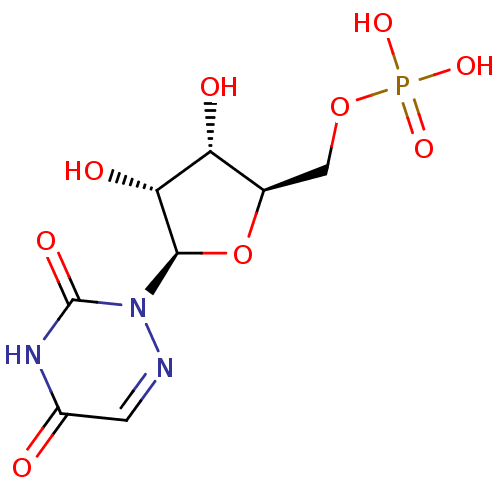 6-aza-UMP {[(2R,3S,4R,5R)-5-(3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-2-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid C6-Uridine Derivative, 18 BDBM21340
6-aza-UMP {[(2R,3S,4R,5R)-5-(3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-2-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid C6-Uridine Derivative, 18 BDBM21340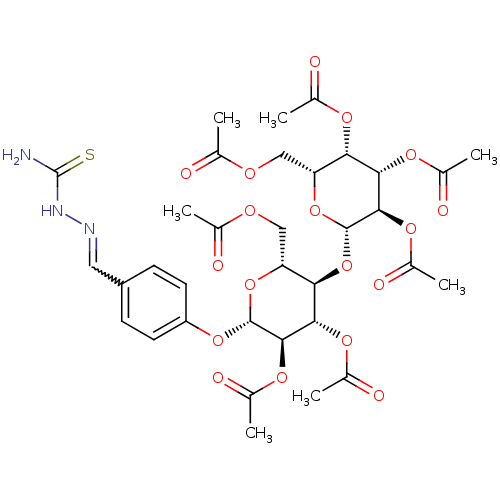 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302745 CHEMBL566863
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302745 CHEMBL566863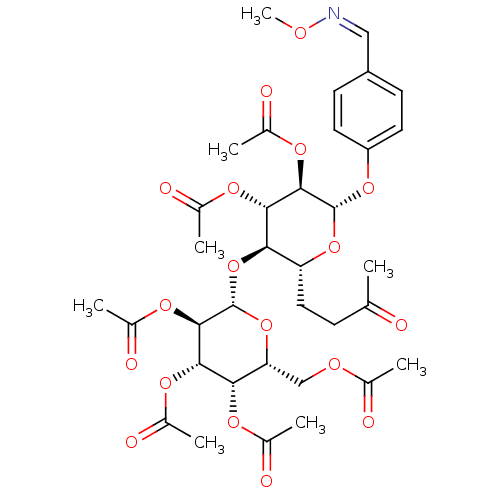 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-6-(4-((methoxyimino)methyl)phenoxy)-2-(3-oxobutyl)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL565507 BDBM50302731
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-6-(4-((methoxyimino)methyl)phenoxy)-2-(3-oxobutyl)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL565507 BDBM50302731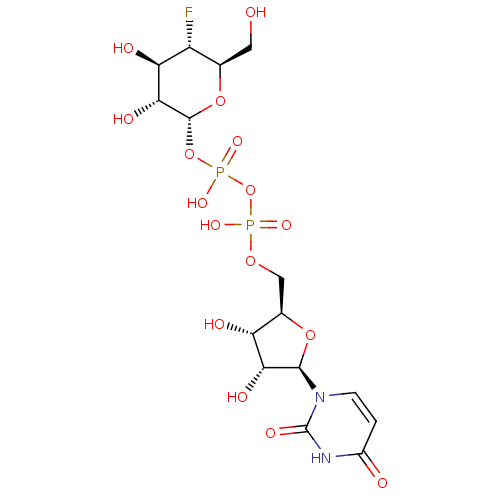 Diphosphoric Acid 1''-alpha-D-[1''](4''-Fluoro-4''-Deoxy)-Glucopyranosyl Ester 2-(uridin-5'-yl)ester CHEMBL593125 URIDINE-5'-DIPHOSPHATE-4-DEOXY-4-FLUORO-ALPHA-D-GALACTOSE BDBM50304034
Diphosphoric Acid 1''-alpha-D-[1''](4''-Fluoro-4''-Deoxy)-Glucopyranosyl Ester 2-(uridin-5'-yl)ester CHEMBL593125 URIDINE-5'-DIPHOSPHATE-4-DEOXY-4-FLUORO-ALPHA-D-GALACTOSE BDBM50304034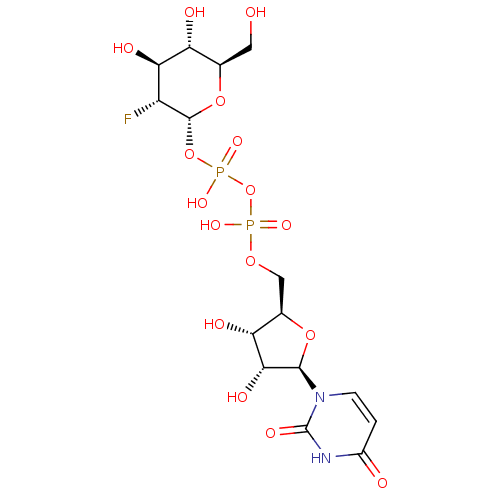 URIDINE-5'-DIPHOSPHATE-2-DEOXY-2-FLUORO-ALPHA-D-GLUCOSE CHEMBL593830 BDBM50304032 Diphosphoric Acid 1''-alpha-D-[1''](2''-Fluoro-2''-Deoxy)Glucopyranosyl Ester 2-(uridin-5'-yl)ester
URIDINE-5'-DIPHOSPHATE-2-DEOXY-2-FLUORO-ALPHA-D-GLUCOSE CHEMBL593830 BDBM50304032 Diphosphoric Acid 1''-alpha-D-[1''](2''-Fluoro-2''-Deoxy)Glucopyranosyl Ester 2-(uridin-5'-yl)ester JMC521648 Compound 34 {[(2R,3S,4R,5R)-5-(6-cyano-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM21335 6-cyanouridine 5-monophosphate C6-Uridine Derivative, 6
JMC521648 Compound 34 {[(2R,3S,4R,5R)-5-(6-cyano-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM21335 6-cyanouridine 5-monophosphate C6-Uridine Derivative, 6 CHEMBL444912 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((1-(4-sulfamoylphenyl)-1H-1,2,3-triazol-4-yl)methylsulfonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50278780 4-(4-{[2',3',4',6'-Tetra-O-acetyl-beta-D-glucopyranosyl]sulfonylmethyl}-1-H-1,2,3-triazol-1-yl)benzenesulfonamide
CHEMBL444912 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((1-(4-sulfamoylphenyl)-1H-1,2,3-triazol-4-yl)methylsulfonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50278780 4-(4-{[2',3',4',6'-Tetra-O-acetyl-beta-D-glucopyranosyl]sulfonylmethyl}-1-H-1,2,3-triazol-1-yl)benzenesulfonamide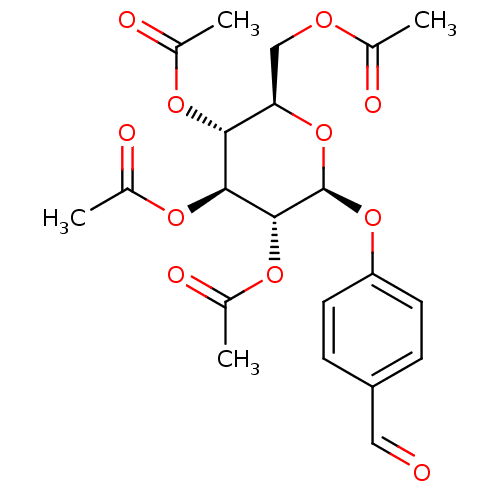 CHEMBL461514 BDBM50245883 Acetic acid (2S,3R,4S,5R,6R)-4,5-diacetoxy-6-acetoxymethyl-2-(4-formyl-phenoxy)-tetrahydro-pyran-3-yl ester (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-glucopyranoside
CHEMBL461514 BDBM50245883 Acetic acid (2S,3R,4S,5R,6R)-4,5-diacetoxy-6-acetoxymethyl-2-(4-formyl-phenoxy)-tetrahydro-pyran-3-yl ester (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-glucopyranoside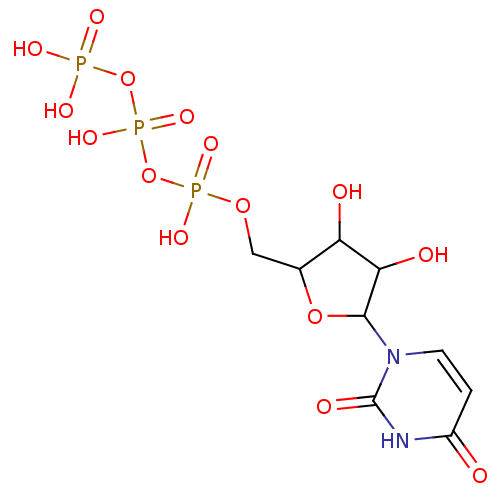 UTP BDBM50023910 5-azidouridine-5'-triphosphate derivative 1-(3,4-dihydroxy-5-triphosphoryloxymethyltetrahydro-2-furanyl)-1,2,3,4-tetrahydro-2,4-pyrimidinedione Uridine triphosphate (UTP) ({[({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)phosphonic acid
UTP BDBM50023910 5-azidouridine-5'-triphosphate derivative 1-(3,4-dihydroxy-5-triphosphoryloxymethyltetrahydro-2-furanyl)-1,2,3,4-tetrahydro-2,4-pyrimidinedione Uridine triphosphate (UTP) ({[({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)phosphonic acid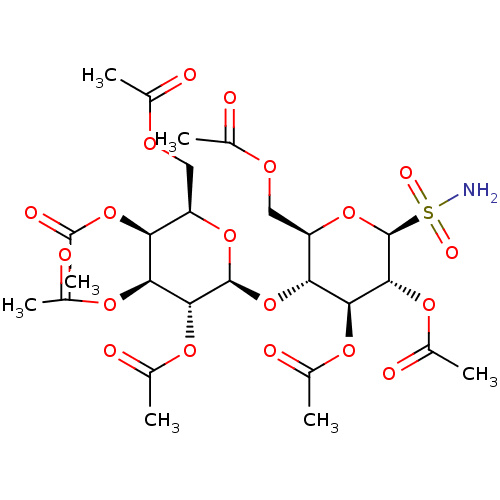 BDBM50299576 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573459 (1S)-2,3,6-tri-O-acetyl-1,5-anhydro-1-sulfamoyl-4-O-(2,3,4,6-tetra-O-acetyl-beta-D-galactopyranosyl)-D-glucitol
BDBM50299576 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573459 (1S)-2,3,6-tri-O-acetyl-1,5-anhydro-1-sulfamoyl-4-O-(2,3,4,6-tetra-O-acetyl-beta-D-galactopyranosyl)-D-glucitol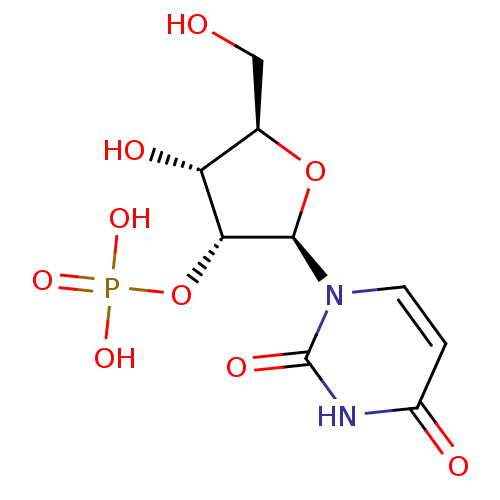 BDBM50292723 uridine 2'-phosphate CHEMBL447360 PHOSPHORIC ACID MONO-[2-(2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YL)-4-HYDROXY-5-HYDROXYMETHYL-TETRAHYDRO-FURAN-3-YL] ESTER (2R,3R,4R,5R)-2-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-3-yl dihydrogen phosphate
BDBM50292723 uridine 2'-phosphate CHEMBL447360 PHOSPHORIC ACID MONO-[2-(2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YL)-4-HYDROXY-5-HYDROXYMETHYL-TETRAHYDRO-FURAN-3-YL] ESTER (2R,3R,4R,5R)-2-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-3-yl dihydrogen phosphate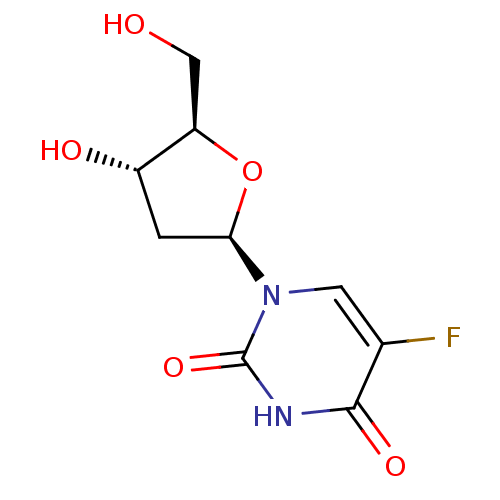 5-fluoro-2'-Deoxy-uridine BDBM50340678 FLOXURIDINE 1-(beta-D-2-deoxy-erythro-pentofuranosyl)-5-fluorouracil 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-fluoro-2'-deoxyuridine 2'-deoxy-5-fluorouridine Fluoruridine Deoxyribose 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione FUDR 5-Fluoro-1-((2R,4S,5R)-4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione 5-Fluoro-1-(4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione CHEMBL917
5-fluoro-2'-Deoxy-uridine BDBM50340678 FLOXURIDINE 1-(beta-D-2-deoxy-erythro-pentofuranosyl)-5-fluorouracil 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-fluoro-2'-deoxyuridine 2'-deoxy-5-fluorouridine Fluoruridine Deoxyribose 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione FUDR 5-Fluoro-1-((2R,4S,5R)-4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione 5-Fluoro-1-(4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione CHEMBL917 (bradykinin triacetate)2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid (BK) H-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg-OH CHEMBL406291 bradykinin BDBM50049949 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid(Bradykinin)
(bradykinin triacetate)2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid (BK) H-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg-OH CHEMBL406291 bradykinin BDBM50049949 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid(Bradykinin)
- Lim, MI; Moyer, JD; Cysyk, RI; Marquez, VE Cyclopentenyluridine and cyclopentenylcytidine analogues as inhibitors of uridine-cytidine kinase. J Med Chem 27: 1536-8 (1985)
- Sun, Q; Yang, Q; Gong, S; Fu, Q; Xiao, Q Synthesis and enzymatic evaluation of phosphoramidon and its� anomer: Anomerization ofa-l-rhamnose triacetate upon phosphitylation. Bioorg Med Chem 21: 6778-87 (2013)
- Pendergast, W; Yerxa, BR; Douglass, JG; Shaver, SR; Dougherty, RW; Redick, CC; Sims, IF; Rideout, JL Synthesis and P2Y receptor activity of a series of uridine dinucleoside 5'-polyphosphates. Bioorg Med Chem Lett 11: 157-60 (2001)
- Van Poecke, S; Barrett, MO; Santhosh Kumar, T; Sinnaeve, D; Martins, JC; Jacobson, KA; Kendall Harden, T; Van Calenbergh, S Synthesis and P2Y2 receptor agonist activities of uridine 5'-phosphonate analogues. Bioorg Med Chem 20: 2304-15 (2012)
- Besada, P; Shin, DH; Costanzi, S; Ko, H; Mathé, C; Gagneron, J; Gosselin, G; Maddileti, S; Harden, TK; Jacobson, KA Structure-activity relationships of uridine 5'-diphosphate analogues at the human P2Y6 receptor. J Med Chem 49: 5532-43 (2006)
- Barb, AW; Leavy, TM; Robins, LI; Guan, Z; Six, DA; Zhou, P; Hangauer, MJ; Bertozzi, CR; Raetz, CR Uridine-based inhibitors as new leads for antibiotics targeting Escherichia coli LpxC. Biochemistry 48: 3068-77 (2009)
- Ko, H; Fricks, I; Ivanov, AA; Harden, TK; Jacobson, KA Structure-activity relationship of uridine 5'-diphosphoglucose analogues as agonists of the human P2Y14 receptor. J Med Chem 50: 2030-9 (2007)
- Bello, AM; Poduch, E; Liu, Y; Wei, L; Crandall, I; Wang, X; Dyanand, C; Kain, KC; Pai, EF; Kotra, LP Structure-activity relationships of C6-uridine derivatives targeting plasmodia orotidine monophosphate decarboxylase. J Med Chem 51: 439-48 (2008)
- Okesli-Armlovich, A; Gupta, A; Jimenez, M; Auld, D; Liu, Q; Bassik, MC; Khosla, C Discovery of small molecule inhibitors of human uridine-cytidine kinase 2 by high-throughput screening. Bioorg Med Chem Lett 29: 2559-2564 (2019)
- Zelikman, V; Pelletier, J; Simhaev, L; Sela, A; Gendron, FP; Arguin, G; Senderowitz, H; S�vigny, J; Fischer, B Highly Selective and Potent Ectonucleotide Pyrophosphatase-1 (NPP1) Inhibitors Based on Uridine 5'-P J Med Chem 61: 3939-3951 (2018)
- Pastuch-Gawolek, G; Plesniak, M; Komor, R; Byczek-Wyrostek, A; Erfurt, K; Szeja, W Synthesis and preliminary biological assay of uridine glycoconjugate derivatives containing amide and/or 1,2,3-triazole linkers. Bioorg Chem 72: 80-88 (2017)
- Hampton, SE; Schipani, A; Bosch-Navarrete, C; Recio, E; Kaiser, M; Kahnberg, P; González-Pacanowska, D; Johansson, NG; Gilbert, IH Investigation of acyclic uridine amide and 5'-amido nucleoside analogues as potential inhibitors of the Plasmodium falciparum dUTPase. Bioorg Med Chem 21: 5876-85 (2013)
- Ko, H; Das, A; Carter, RL; Fricks, IP; Zhou, Y; Ivanov, AA; Melman, A; Joshi, BV; Kovác, P; Hajduch, J; Kirk, KL; Harden, TK; Jacobson, KA Molecular recognition in the P2Y(14) receptor: Probing the structurally permissive terminal sugar moiety of uridine-5'-diphosphoglucose. Bioorg Med Chem 17: 5298-311 (2009)
- Brunschweiger, A; Iqbal, J; Umbach, F; Scheiff, AB; Munkonda, MN; Sévigny, J; Knowles, AF; Müller, CE Selective nucleoside triphosphate diphosphohydrolase-2 (NTPDase2) inhibitors: nucleotide mimetics derived from uridine-5'-carboxamide. J Med Chem 51: 4518-28 (2008)
- Wang, G; Lim, SP; Chen, YL; Hunziker, J; Rao, R; Gu, F; Seh, CC; Ghafar, NA; Xu, H; Chan, K; Lin, X; Saunders, OL; Fenaux, M; Zhong, W; Shi, PY; Yokokawa, F Structure-activity relationship of uridine-based nucleoside phosphoramidate prodrugs for inhibition of dengue virus RNA-dependent RNA polymerase. Bioorg Med Chem Lett 28: 2324-2327 (2018)
- Ii, K; Ichikawa, S; Al-Dabbagh, B; Bouhss, A; Matsuda, A Function-oriented synthesis of simplified caprazamycins: discovery of oxazolidine-containing uridine derivatives as antibacterial agents against drug-resistant bacteria. J Med Chem 53: 3793-813 (2010)
- Das, A; Ko, H; Burianek, LE; Barrett, MO; Harden, TK; Jacobson, KA Human P2Y(14) receptor agonists: truncation of the hexose moiety of uridine-5'-diphosphoglucose and its replacement with alkyl and aryl groups. J Med Chem 53: 471-80 (2010)
- Jayasekara, PS; Barrett, MO; Ball, CB; Brown, KA; Hammes, E; Balasubramanian, R; Harden, TK; Jacobson, KA 4-Alkyloxyimino derivatives of uridine-5'-triphosphate: distal modification of potent agonists as a strategy for molecular probes of P2Y2, P2Y4, and P2Y6 receptors. J Med Chem 57: 3874-83 (2014)
- Renck, D; Machado, P; Souto, AA; Rosado, LA; Erig, T; Campos, MM; Farias, CB; Roesler, R; Timmers, LF; de Souza, ON; Santos, DS; Basso, LA Design of novel potent inhibitors of human uridine phosphorylase-1: synthesis, inhibition studies, thermodynamics, and in vitro influence on 5-fluorouracil cytotoxicity. J Med Chem 56: 8892-902 (2013)
- Orr, GF; Musso, DL; Boswell, GE; Kelley, JL; Joyner, SS; Davis, ST; Baccanari, DP Inhibition of uridine phosphorylase: synthesis and structure-activity relationships of aryl-substituted 5-benzyluracils and 1-[(2-hydroxyethoxy)methyl]-5-benzyluracils. J Med Chem 38: 3850-6 (1995)
- Wnuk, SF; Lewandowska, E; Sacasa, PR; Crain, LN; Zhang, J; Borchardt, RT; De Clercq, E Stereoselective synthesis of sugar-modified enyne analogues of adenosine and uridine. Interaction with S-adenosyl-L-homocysteine hydrolase and antiviral and cytotoxic effects. J Med Chem 47: 5251-7 (2004)
- Wnuk, SF; Ro, BO; Valdez, CA; Lewandowska, E; Valdez, NX; Sacasa, PR; Yin, D; Zhang, J; Borchardt, RT; De Clercq, E Sugar-modified conjugated diene analogues of adenosine and uridine: synthesis, interaction with S-adenosyl-L-homocysteine hydrolase, and antiviral and cytostatic effects. J Med Chem 45: 2651-8 (2002)
- Ohno, K; Sugiyama, D; Takeshita, L; Kanamori, T; Masaki, Y; Sekine, M; Seio, K Synthesis of photocaged 6-O-(2-nitrobenzyl)guanosine and 4-O-(2-nitrobenzyl) uridine triphosphates for photocontrol of the RNA transcription reaction. Bioorg Med Chem 25: 6007-6015 (2017)
- Durrant, JD; Urbaniak, MD; Ferguson, MA; McCammon, JA Computer-aided identification of Trypanosoma brucei uridine diphosphate galactose 4'-epimerase inhibitors: toward the development of novel therapies for African sleeping sickness. J Med Chem 53: 5025-32 (2010)
- Alexandre, FR; Badaroux, E; Bilello, JP; Bot, S; Bouisset, T; Brandt, G; Cappelle, S; Chapron, C; Chaves, D; Convard, T; Counor, C; Da Costa, D; Dukhan, D; Gay, M; Gosselin, G; Griffon, JF; Gupta, K; Hernandez-Santiago, B; La Colla, M; Lioure, MP; Milhau, J; Paparin, JL; Peyronnet, J; Parsy, C; Pierra Rouvi�re, C; Rahali, H; Rahali, R; Salanson, A; Seifer, M; Serra, I; Standring, D; Surleraux, D; Dousson, CB The discovery of IDX21437: Design, synthesis and antiviral evaluation of 2'-?-chloro-2'-?-C-methyl branched uridine pronucleotides as potent liver-targeted HCV polymerase inhibitors. Bioorg Med Chem Lett 27: 4323-4330 (2017)
- Wang, G; Dyatkina, N; Prhavc, M; Williams, C; Serebryany, V; Hu, Y; Huang, Y; Wan, J; Wu, X; Deval, J; Fung, A; Jin, Z; Tan, H; Shaw, K; Kang, H; Zhang, Q; Tam, Y; Stoycheva, A; Jekle, A; Smith, DB; Beigelman, L Synthesis and Anti-HCV Activities of 4'-Fluoro-2'-Substituted Uridine Triphosphates and Nucleotide Prodrugs: Discovery of 4'-Fluoro-2'- C-methyluridine 5'-Phosphoramidate Prodrug (AL-335) for the Treatment of Hepatitis C Infection. J Med Chem 62: 4555-4570 (2019)
- Jonckers, TH; Tahri, A; Vijgen, L; Berke, JM; Lachau-Durand, S; Stoops, B; Snoeys, J; Leclercq, L; Tambuyzer, L; Lin, TI; Simmen, K; Raboisson, P Discovery of 1-((2R,4aR,6R,7R,7aR)-2-Isopropoxy-2-oxidodihydro-4H,6H-spiro[furo[3,2-d][1,3,2]dioxaphosphinine-7,2'-oxetan]-6-yl)pyrimidine-2,4(1H,3H)-dione (JNJ-54257099), a 3'-5'-Cyclic Phosphate Ester Prodrug of 2'-Deoxy-2'-Spirooxetane Uridine Triphosphate Useful for HCV Inhibition. J Med Chem 59: 5790-8 (2016)
- ChEMBL_684504 (CHEMBL1287513) Inhibition of human uridine phosphorylase
- ChEMBL_212951 (CHEMBL873996) Inhibition of uridine phosphorylase (UrdPase) from murine liver.
- ChEMBL_639016 (CHEMBL1166154) Inhibition of Saccharomyces cerevisia uridine 5'-monophosphate synthase
- ChEMBL_2273267 Inhibition of human recombinant ENT1 assessed as inhibition of [3H] uridine transport
- ChEBML_212946 Binding affinity to uridine kinase from L1210. was determined from the dixon plot
- ChEBML_212947 Binding affinity to uridine kinase from L1210. was determined from the dixon plot
- ChEMBL_1337494 (CHEMBL3241962) Inhibition of human recombinant cytidine deaminase assessed as cytidine to uridine formation
- ChEMBL_518728 (CHEMBL957103) Binding affinity to human ENT1 assessed as [3H]uridine uptake by flow cytometry
- ChEMBL_472344 (CHEMBL952147) Inhibition of glucan synthase in Candida albicans 36082 assessed as incorporation of [3H]uridine diphosphoglucose
- ChEMBL_639005 (CHEMBL1168988) Inhibition of human uridine 5'-monophosphate synthase after overnight incubation at room temperature by UV spectroscopy
- ChEMBL_1432010 (CHEMBL3388115) Inhibition of human ENT1 expressed in porcine PK15NTD cells by cell-based [3H]5-uridine uptake assay
- ChEMBL_1432011 (CHEMBL3388116) Inhibition of rat ENT2 expressed in rat H9c2 cells by cell-based 3H[5-]uridine uptake assay
- ChEMBL_472345 (CHEMBL952148) Inhibition of glucan synthase in Candida albicans 36082 assessed as incorporation of [3H]uridine diphosphoglucose in presence of 10% serum
- ChEMBL_472346 (CHEMBL952149) Inhibition of glucan synthase in Candida albicans 36082 assessed as incorporation of [3H]uridine diphosphoglucose in presence of 20% serum
- ChEMBL_472347 (CHEMBL952150) Inhibition of glucan synthase in Candida albicans 36082 assessed as incorporation of [3H]uridine diphosphoglucose in presence of 50% serum
- ChEMBL_212952 (CHEMBL816766) The compound was tested for inhibition of uridine phosphorylase (UrdPase) from murine liver Value refers to activity for apparent Ki value
- ChEMBL_639011 (CHEMBL1169125) Inhibition of Plasmodium falciparum uridine 5'-monophosphate synthase after overnight incubation at room temperature by VP-ITC microcalorimetry
- ChEMBL_639015 (CHEMBL1166153) Inhibition of Saccharomyces cerevisia uridine 5'-monophosphate synthase after overnight incubation at room temperature by VP-ITC microcalorimetry
- ChEMBL_518727 (CHEMBL957102) Binding affinity to human recombinant CNT3 expressed in pig PK15NTD cells assessed as [3H]uridine uptake by beta-scintillation counter
- ChEMBL_1494513 (CHEMBL3528990) Binding affinity to recombinant human ENT1 expressed in Saccharomyces cerevisiae assessed as inhibition of [3H]-uridine transport after 15 mins by scintillation counting analysis
- ChEMBL_1494514 (CHEMBL3528991) Binding affinity to recombinant human ENT2 expressed in Saccharomyces cerevisiae assessed as inhibition of [3H]-uridine transport after 15 mins by scintillation counting analysis
- ChEMBL_1494515 (CHEMBL3528992) Binding affinity to recombinant human CNT1 expressed in Saccharomyces cerevisiae assessed as inhibition of [3H]-uridine transport after 15 mins by scintillation counting analysis
- ChEMBL_1494516 (CHEMBL3528993) Binding affinity to recombinant human CNT2 expressed in Saccharomyces cerevisiae assessed as inhibition of [3H]-uridine transport after 15 mins by scintillation counting analysis
- ChEMBL_1494517 (CHEMBL3528994) Binding affinity to recombinant human CNT3 expressed in Saccharomyces cerevisiae assessed as inhibition of [3H]-uridine transport after 5 mins by scintillation counting analysis
- ChEMBL_1341180 (CHEMBL3256792) Competitive inhibition of thymidylate synthase purified from methotrexate-resistant Lactobacillus casei using 2'-deoxy[5-3H]uridine 5'-phosphate by radioisotope assay
- ChEMBL_2261597 (CHEMBL5216608) Inhibition of human ENT1 transfected in nucleoside transporter-deficient pig PK-15 cells assessed as inhibition of [3H]-uridine uptake by liquid scintillation counter analysis
- ChEMBL_2261598 (CHEMBL5216609) Inhibition of human ENT2 transfected in nucleoside transporter-deficient pig PK-15 cells assessed as inhibition of [3H]-uridine uptake by liquid scintillation counter analysis
- ChEMBL_1589257 (CHEMBL3830285) Inhibition of Crithidia fasciculata inosine-uridine nucleoside hydrolase expressed in Escherichia coli using p-nitrophenyl beta-D-ribofuranoside as substrate by xanthine oxidase coupled enzyme assay
- ChEMBL_1333129 (CHEMBL3232284) Competitive inhibition of Lactobacillus casei thymidylate synthetase using 2'-deoxy[5-3H]uridine-5'-phosphate as substrate assessed as release of water after 15 mins by double reciprocal plot analysis
- Enzyme Assay The aim of this study was to evaluate TK-112690 in vivo as an inhibitor of uridine phosphorylase (UPase) enzyme activity. The range of TK-112690 doses studied for ability to prevent metabolic breakdown of uridine, through the in vitro inhibition of mouse and human small intestinal UPase enzyme, was 0, 0.1, 0.5, 1, 5, 10, 50, 100, 500, 1000, 5000 and 10000 uM). Detection of UPase activity was determined by HPLC analysis using UV detection of uracil concentration (UPase catabolizes uridine into uracil and ribose-1-phosphate).The UPase enzyme material was prepared from homogenized mouse and human being small intestinal tissue. TK-112690 was dissolved in water (50 mg/ml) and analyzed for UPase inhibition in aqueous solution containing 5 mM uridine, 0.01 M Tris, 0.01 M phosphate, 1 mM EDTA, and 1 mM DTT. Reactions were performed at 37 C. at pH of 7.3.TK-11260 inhibition of mouse and human UPase was analyzed by reverse phase HPLC using UV detection.
- ChEMBL_1886104 (CHEMBL4387686) Non-competitive inhibition of C-terminally His6-tagged human UCK2 expressed in Escherichia coli BL21(DE3) cells using phosphoenolpyruvate, NADH, uridine level by spectrophotometry based pyruvate kinase and lactate dehydrogenase coupled enzyme assay
- ChEMBL_1886100 (CHEMBL4387682) Non-competitive inhibition of C-terminally His6-tagged human UCK2 expressed in Escherichia coli BL21(DE3) cells using phosphoenolpyruvate, NADH and varying uridine level by spectrophotometry based pyruvate kinase and lactate dehydrogenase coupled enzyme assay
- ChEMBL_1886101 (CHEMBL4387683) Non-competitive inhibition of C-terminally His6-tagged human UCK2 expressed in Escherichia coli BL21(DE3) cells using uridine, phosphoenolpyruvate, NADH and varying ATP level by spectrophotometry based pyruvate kinase and lactate dehydrogenase coupled enzyme assay
- Study with Mouse and Human Intestinal Tissue Homogenates The aim of this study was to evaluate TK-112690 in vivo as an inhibitor of uridine phosphorylase (UPase) enzyme activity. The range of TK-112690 doses studied for ability to prevent metabolic breakdown of uridine, through the in vitro inhibition of mouse and human small intestinal UPase enzyme, was 0, 0.1, 0.5, 1, 5, 10, 50, 100, 500, 1000, 5000 and 10000 μM). Detection of UPase activity was determined by HPLC analysis using UV detection of uracil concentration (UPase catabolizes uridine into uracil and ribose-1-phosphate).The UPase enzyme material was prepared from homogenized mouse and human being small intestinal tissue. TK-112690 was dissolved in water (50 mg/ml) and analyzed for UPase inhibition in aqueous solution containing 5 mM uridine, 0.01 M Tris, 0.01 M phosphate, 1 mM EDTA, and 1 mM DTT. Reactions were performed at 37� C. at pH of 7.3.TK-11260 inhibition of mouse and human UPase was analyzed by reverse phase HPLC using UV detection. HPLC analysis was performed at ambient temperature with a Water 2695 Alliance system equipped with a C18 ECONOSIL 5 U ALLtech column. 20 μl of UPase reaction samples were auto-injected onto column. Mobile phase consisted of water eluting for first 2.5 ml and acetonitrile gradient to 100% in 12.5 ml (flow rate 0.5 ml/min). The outlet fluent was monitored by UV absorption in the range of 240-320 nm. UPase enzymatic activity was based on the AUC of the uracil peaks.
- P. aeruginosa MurC assay The reactions (50 μL) were carried out in 50 mM Tris-HCl pH 8.0, 20 mM ammonium sulfate, 2.5 mM DTT, 0.002% Brij-35, 1 mM MgCl2, 18 μM UNAM, 38 μM ATP, 28 μM L-alanine, 2 μg/mL Poly uridine nucleic acid, 0.1 μM Polynucleotide Phosphorylase, 0.25X ribogreen and 3 nM enzyme in 384-well plate at 25 C for 60 minutes.
- Enzyme Assay The procedure to determine CDA enzymatic activity is based on published methodologies (for example, Cacciamani, T. et al., Arch. Biochem. Biophys. 1991, 290, 285-92; Cohen R. et al., J. Biol. Chem., 1971, 246, 7566-8; Vincenzetti S. et al., Protein Expr. Purif. 1996, 8, 247-53). The assay follows the change in absorbance at 286 nm of the CDA-catalyzed deamination of cytidine to form uridine. The reaction is carried out in potassium phosphate buffer (pH 7.4, 20 mM, containing 1mM DTT) in a total volume of 200 μl in a 96-well plate format. The final reaction mixture contains cytidine (50 μM) and purified human recombinant CDA. Purified enzyme is diluted so as to produce an absorbance change of approximately 2 milli-absorbance units/minute. Base line measurements of absorbance change over time are made before CDA addition to insure no change of absorbance in the absence of CDA. After CDA addition, absorbance change is monitored for 20-30 minutes.
 Calyculin A 11,13,21-triacetate (12) BDBM40779
Calyculin A 11,13,21-triacetate (12) BDBM40779 Jaceidin triacetate BDBM645366 US20240016777, Table3.8
Jaceidin triacetate BDBM645366 US20240016777, Table3.8 CHEMBL130266 UDP uridine 5'-(trihydrogen diphosphate) BDBM50118239 Uridine diphosphate
CHEMBL130266 UDP uridine 5'-(trihydrogen diphosphate) BDBM50118239 Uridine diphosphate BDBM50088517 Uridine CHEBI:16704
BDBM50088517 Uridine CHEBI:16704 UDPG BDBM50423218 URIDINE DIPHOSPHATE GLUCOSE Udp-Glucose 5''-Diphosphoglucose Uridine-5''-Diphosphoglucose
UDPG BDBM50423218 URIDINE DIPHOSPHATE GLUCOSE Udp-Glucose 5''-Diphosphoglucose Uridine-5''-Diphosphoglucose BDBM50118213 UTP uridine 5'-triphosphoric acid H4utp CHEMBL336296 uridine 5'-(tetrahydrogen triphosphate) 5'-UTP
BDBM50118213 UTP uridine 5'-triphosphoric acid H4utp CHEMBL336296 uridine 5'-(tetrahydrogen triphosphate) 5'-UTP BDBM50173721 Uridine-5'-diphosphogalactose derivative CHEMBL196432
BDBM50173721 Uridine-5'-diphosphogalactose derivative CHEMBL196432 BDBM50271289 Uridine-5'-methyl-tetraphosphate CHEMBL519991
BDBM50271289 Uridine-5'-methyl-tetraphosphate CHEMBL519991 CHEMBL1096401 BDBM50318022 uridine diphosphate trisodium salt
CHEMBL1096401 BDBM50318022 uridine diphosphate trisodium salt CHEMBL227711 BDBM50209666 URIDINE-5'-DIPHOSPHATE-MANNOSE
CHEMBL227711 BDBM50209666 URIDINE-5'-DIPHOSPHATE-MANNOSE CHEMBL474887 BDBM50270542 Uridine-5'-cyclohexane-tetraphosphate
CHEMBL474887 BDBM50270542 Uridine-5'-cyclohexane-tetraphosphate Galactose-Uridine-5'-Diphosphate CHEMBL439009 BDBM50209668
Galactose-Uridine-5'-Diphosphate CHEMBL439009 BDBM50209668 Uridine-5'-phenyl-tetraphosphate BDBM50270541 CHEMBL474886
Uridine-5'-phenyl-tetraphosphate BDBM50270541 CHEMBL474886 BDBM50205418 CHEMBL410594 uridine 5'-tetraphosphate 5'-ribose
BDBM50205418 CHEMBL410594 uridine 5'-tetraphosphate 5'-ribose BDBM50271252 2-Thio-uridine-5'-tetraphosphate CHEMBL482485
BDBM50271252 2-Thio-uridine-5'-tetraphosphate CHEMBL482485 BDBM50271254 CHEMBL482683 2-Thio-uridine-5'-pentaphosphate
BDBM50271254 CHEMBL482683 2-Thio-uridine-5'-pentaphosphate BDBM50345487 Uridine-5'-(4-chlorophenyl)-tetraphosphate CHEMBL1784895
BDBM50345487 Uridine-5'-(4-chlorophenyl)-tetraphosphate CHEMBL1784895 BDBM50345491 Uridine-5'-allose-1'-tetraphosphate CHEMBL1784885
BDBM50345491 Uridine-5'-allose-1'-tetraphosphate CHEMBL1784885 BDBM50345493 CHEMBL1784887 Uridine-5'-xylose-1'-tetraphosphate
BDBM50345493 CHEMBL1784887 Uridine-5'-xylose-1'-tetraphosphate CHEMBL1784900 Uridine-5'-(3-nitrophenyl)-tetraphosphate BDBM50345490
CHEMBL1784900 Uridine-5'-(3-nitrophenyl)-tetraphosphate BDBM50345490 CHEMBL500840 BDBM50270544 Uridine-5'-fructose-6'-tetraphosphate
CHEMBL500840 BDBM50270544 Uridine-5'-fructose-6'-tetraphosphate CHEMBL508122 Uridine-5'-mannose-6'-tetraphosphate BDBM50270547
CHEMBL508122 Uridine-5'-mannose-6'-tetraphosphate BDBM50270547 CHEMBL521451 BDBM50271253 4-Thio-uridine-5'-tetraphosphate
CHEMBL521451 BDBM50271253 4-Thio-uridine-5'-tetraphosphate Uridine-5'-(2-cyanoethyl)-tetraphosphate BDBM50271291 CHEMBL484105
Uridine-5'-(2-cyanoethyl)-tetraphosphate BDBM50271291 CHEMBL484105 Uridine-5'-(3-chlorophenyl)-tetraphosphate BDBM50345488 CHEMBL1784896
Uridine-5'-(3-chlorophenyl)-tetraphosphate BDBM50345488 CHEMBL1784896 Uridine-5'-(4-nitrophenyl)-tetraphosphate CHEMBL1784899 BDBM50345489
Uridine-5'-(4-nitrophenyl)-tetraphosphate CHEMBL1784899 BDBM50345489 Uridine-5'-alpha-glycerol-tetraphosphate CHEMBL482895 BDBM50271292
Uridine-5'-alpha-glycerol-tetraphosphate CHEMBL482895 BDBM50271292 Uridine-5'-galactose-1'-tetraphosphate BDBM50270546 CHEMBL444212
Uridine-5'-galactose-1'-tetraphosphate BDBM50270546 CHEMBL444212 Uridine-5'-glucose-1'-tetraphosphate BDBM50270545 CHEMBL499138
Uridine-5'-glucose-1'-tetraphosphate BDBM50270545 CHEMBL499138 Uridine-5'-mannose-1'-tetraphosphate BDBM50345492 CHEMBL1784886
Uridine-5'-mannose-1'-tetraphosphate BDBM50345492 CHEMBL1784886 CHEMBL228057 uridine 5'-[3-(alpha-D-glucopyranuronosyl) dihydrogen diphosphate] BDBM50209665 uridine diphosphate glucuronic acid UDP-alpha-D-glucuronic acid
CHEMBL228057 uridine 5'-[3-(alpha-D-glucopyranuronosyl) dihydrogen diphosphate] BDBM50209665 uridine diphosphate glucuronic acid UDP-alpha-D-glucuronic acid CHEMBL483913 Uridine-5'-methyl(C-P)-tetraphosphate BDBM50271290
CHEMBL483913 Uridine-5'-methyl(C-P)-tetraphosphate BDBM50271290 CHEMBL521487 BDBM50271224 2'-C-Methyl-uridine-5'-triphosphate
CHEMBL521487 BDBM50271224 2'-C-Methyl-uridine-5'-triphosphate P1-Uridine 5'-P3-Cyclohexyltriphosphate TriethylammoniumSalt CHEMBL1083258 BDBM50319138
P1-Uridine 5'-P3-Cyclohexyltriphosphate TriethylammoniumSalt CHEMBL1083258 BDBM50319138 CHEMBL451438 cholest-8-ene-3beta,5alpha,6alpha,7alpha,10alpha-pentol 3,6,7-triacetate BDBM50269197
CHEMBL451438 cholest-8-ene-3beta,5alpha,6alpha,7alpha,10alpha-pentol 3,6,7-triacetate BDBM50269197 5'-deoxy-5'-N-(ethyl isonipecotatyl)uridine BDBM50292715 CHEMBL445773
5'-deoxy-5'-N-(ethyl isonipecotatyl)uridine BDBM50292715 CHEMBL445773 BDBM50271188 CHEMBL482472 2-Thio-4-methylthio-uridine-5'-triphosphate
BDBM50271188 CHEMBL482472 2-Thio-4-methylthio-uridine-5'-triphosphate BDBM50342006 5'-Deoxy-5'-N-(4-carboxypiperidinyl)uridine CHEMBL1765476
BDBM50342006 5'-Deoxy-5'-N-(4-carboxypiperidinyl)uridine CHEMBL1765476 CHEMBL1083259 BDBM50319137 P1-Uridine 5'-P3-Phenyltriphosphate Triethylammonium Salt
CHEMBL1083259 BDBM50319137 P1-Uridine 5'-P3-Phenyltriphosphate Triethylammonium Salt CHEMBL454230 Uridine-5'-(2'-deoxy-glucose)-6'-tetraphosphate BDBM50270548
CHEMBL454230 Uridine-5'-(2'-deoxy-glucose)-6'-tetraphosphate BDBM50270548 hydrogen ({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl phosphonato}oxy)phosphonate BDBM50096295 Uridine dinucleoside 5'-polyphosphate analogue Uridine
hydrogen ({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl phosphonato}oxy)phosphonate BDBM50096295 Uridine dinucleoside 5'-polyphosphate analogue Uridine 3'-[1-(4-ethoxycarbonylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL471836 BDBM50276040
3'-[1-(4-ethoxycarbonylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL471836 BDBM50276040 BDBM50270551 P1,P4-di(uridine 5'-)beta,gamma-difluoromethylenetetraphosphate CHEMBL502712
BDBM50270551 P1,P4-di(uridine 5'-)beta,gamma-difluoromethylenetetraphosphate CHEMBL502712 BDBM50292714 CHEMBL461790 3'-N-piperidine-4-carboxyl-3'-deoxyara-uridine
BDBM50292714 CHEMBL461790 3'-N-piperidine-4-carboxyl-3'-deoxyara-uridine BDBM50319134 Uridine 5'-Glucose-1'-alpha,beta-methylenetriphosphate TriethylammoniumSalt CHEMBL1083263
BDBM50319134 Uridine 5'-Glucose-1'-alpha,beta-methylenetriphosphate TriethylammoniumSalt CHEMBL1083263 CHEMBL1784902 BDBM50345495 P1-(Uridine-5'-)P4-(glucuronic acid-1'-)tetraphosphate
CHEMBL1784902 BDBM50345495 P1-(Uridine-5'-)P4-(glucuronic acid-1'-)tetraphosphate P1,P4-di(uridine 5'-)beta,gamma-dichloromethlyenetetraphosphate BDBM50270552 CHEMBL476021
P1,P4-di(uridine 5'-)beta,gamma-dichloromethlyenetetraphosphate BDBM50270552 CHEMBL476021 P1-Uridine 5'-P3-[1]Glucose-1'-Triphosphate TriethylammoniumSalt BDBM50319132 CHEMBL1083260
P1-Uridine 5'-P3-[1]Glucose-1'-Triphosphate TriethylammoniumSalt BDBM50319132 CHEMBL1083260 (3R,4R,5R,6R)-3-acetamido-6-(acetoxymethyl)tetrahydro-2H-pyran-2,4,5-triyl triacetate CHEMBL610736 BDBM50304486
(3R,4R,5R,6R)-3-acetamido-6-(acetoxymethyl)tetrahydro-2H-pyran-2,4,5-triyl triacetate CHEMBL610736 BDBM50304486 BDBM50299574 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL579226
BDBM50299574 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL579226 BDBM50299575 (2S,3S,4S,5R,6S)-2-(methoxycarbonyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL584312
BDBM50299575 (2S,3S,4S,5R,6S)-2-(methoxycarbonyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL584312 CHEMBL568705 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50299573
CHEMBL568705 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50299573 BDBM50345496 P1-(Uridine-5'-)P4-(3'-deoxy-3'-fluoroglucose-1'-)tetraphosphate CHEMBL1784904
BDBM50345496 P1-(Uridine-5'-)P4-(3'-deoxy-3'-fluoroglucose-1'-)tetraphosphate CHEMBL1784904 CHEMBL1784293 P1-(Uridine-5'-)P4-(4'-deoxy-4'-fluoroglucose-1'-)tetraphosphate BDBM50345499
CHEMBL1784293 P1-(Uridine-5'-)P4-(4'-deoxy-4'-fluoroglucose-1'-)tetraphosphate BDBM50345499 P1-(Uridine-5'-)P4-(2'-deoxy-2'-acetamidoglucose-1'-)tetraphosphate CHEMBL1784889 BDBM50345494
P1-(Uridine-5'-)P4-(2'-deoxy-2'-acetamidoglucose-1'-)tetraphosphate CHEMBL1784889 BDBM50345494 CHEMBL1782243 7-methoxy-2,8-bis(3-methylbut-2-enyl)-9-oxo-9H-xanthene-1,3,6-triyl triacetate BDBM50346338
CHEMBL1782243 7-methoxy-2,8-bis(3-methylbut-2-enyl)-9-oxo-9H-xanthene-1,3,6-triyl triacetate BDBM50346338 CHEMBL44247 Acetic acid 3,8-diacetoxy-6-methyl-9,10-dioxo-9,10-dihydro-anthracen-1-yl ester emodin triacetate BDBM50005914
CHEMBL44247 Acetic acid 3,8-diacetoxy-6-methyl-9,10-dioxo-9,10-dihydro-anthracen-1-yl ester emodin triacetate BDBM50005914 uridine 5'-[3-alpha-D-glucopyranosyl dihydrogen diphosphate] UDP-alpha-D-glucose BDBM50209659
uridine 5'-[3-alpha-D-glucopyranosyl dihydrogen diphosphate] UDP-alpha-D-glucose BDBM50209659 BDBM50341904 CHEMBL1767407 [P(1)-(uridine 5')-P(4)-(2'-deoxycytidine 5')tetraphosphate,tetrasodium salt]
BDBM50341904 CHEMBL1767407 [P(1)-(uridine 5')-P(4)-(2'-deoxycytidine 5')tetraphosphate,tetrasodium salt] (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302735 CHEMBL583541
(2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302735 CHEMBL583541 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302741 CHEMBL569784
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302741 CHEMBL569784 BDBM50302742 CHEMBL569314 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
BDBM50302742 CHEMBL569314 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL570195 BDBM50302734 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
CHEMBL570195 BDBM50302734 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL571325 BDBM50302740 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate
CHEMBL571325 BDBM50302740 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50303334 Diphosphoric Acid 1-r-Glycerol Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303334 Diphosphoric Acid 1-r-Glycerol Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303335 Diphosphoric Acid 1-beta-Glycerol Ester 2-(Uridine-5'-yl)ester,bis-Triethylammonium Salt
BDBM50303335 Diphosphoric Acid 1-beta-Glycerol Ester 2-(Uridine-5'-yl)ester,bis-Triethylammonium Salt BDBM50303336 Diphosphoric Acid 1-beta-Cyclohexyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303336 Diphosphoric Acid 1-beta-Cyclohexyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303348 Diphosphoric Acid 1-beta-Phenyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303348 Diphosphoric Acid 1-beta-Phenyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50304031 Diphosphoric Acid 1''-alpha-D-[1'']Fucopyranosyl Ester 2-(uridine-5'-yl)ester CHEMBL595940
BDBM50304031 Diphosphoric Acid 1''-alpha-D-[1'']Fucopyranosyl Ester 2-(uridine-5'-yl)ester CHEMBL595940 BDBM50341905 CHEMBL1765124 ammonium 6-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate
BDBM50341905 CHEMBL1765124 ammonium 6-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341906 Ammonium 6-Amido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate CHEMBL1765121
BDBM50341906 Ammonium 6-Amido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate CHEMBL1765121 CHEMBL1765126 ammonium 6-Azido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341909
CHEMBL1765126 ammonium 6-Azido-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341909 Diphosphoric Acid 1-beta-Methyl Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303341
Diphosphoric Acid 1-beta-Methyl Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303341 P1-((2-benzyl-1,3-dioxolo-4-yl)uridine 5')P3-(5-iodouridine5') triphosphate CHEMBL503256 BDBM50270549
P1-((2-benzyl-1,3-dioxolo-4-yl)uridine 5')P3-(5-iodouridine5') triphosphate CHEMBL503256 BDBM50270549 ammonium 5-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341907 CHEMBL1765122
ammonium 5-Cyano-2'-deoxy-2'-fluoro-beta-D-uridine 5'-O-Monophosphate BDBM50341907 CHEMBL1765122 BDBM21338 6-Amino-uridine 5-O-Monophosphate C6-Uridine Derivative, 15 JMC521648 Compound 37 {[(2R,3S,4R,5R)-5-(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
BDBM21338 6-Amino-uridine 5-O-Monophosphate C6-Uridine Derivative, 15 JMC521648 Compound 37 {[(2R,3S,4R,5R)-5-(6-amino-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid JMC521648 Compound 36 C6-Uridine Derivative, 14 6-Azido-uridine 5-O-Monophosphate BDBM21337 {[(2R,3S,4R,5R)-5-(6-azido-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
JMC521648 Compound 36 C6-Uridine Derivative, 14 6-Azido-uridine 5-O-Monophosphate BDBM21337 {[(2R,3S,4R,5R)-5-(6-azido-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302749 CHEMBL569974
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302749 CHEMBL569974 BDBM50302748 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL569079
BDBM50302748 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL569079 BDBM50302750 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL568781
BDBM50302750 (2R,3R,4R,5R,6S)-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL568781 BDBM21339 C6-Uridine Derivative, 16 {[(2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)oxolan-2-yl]methoxy}phosphonic acid JMC521648 Compound 38 6-Methyl-uridine 5-O-Monophosphate
BDBM21339 C6-Uridine Derivative, 16 {[(2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)oxolan-2-yl]methoxy}phosphonic acid JMC521648 Compound 38 6-Methyl-uridine 5-O-Monophosphate BDBM50303332 Diphosphoric Acid 1-beta-(2-Cyanoethyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt
BDBM50303332 Diphosphoric Acid 1-beta-(2-Cyanoethyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303337 Diphosphoric Acid 1-beta-(4-Methoxyphenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303337 Diphosphoric Acid 1-beta-(4-Methoxyphenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303349 Diphosphoric Acid 1-beta-(4-Nitrophenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt
BDBM50303349 Diphosphoric Acid 1-beta-(4-Nitrophenyl)Ester 2-(Uridine-5'-yl)ester,di ammonium Salt Diphosphoric Acid 1-beta-(3-Butynyl)Ester 2-(Uridine-5'-yl)-ester, bis-Triethylammonium Salt BDBM50303345
Diphosphoric Acid 1-beta-(3-Butynyl)Ester 2-(Uridine-5'-yl)-ester, bis-Triethylammonium Salt BDBM50303345 Diphosphoric Acid 1-beta-(3-Chlorophenyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303350
Diphosphoric Acid 1-beta-(3-Chlorophenyl)Ester 2-(Uridine-5'-yl)ester, di ammonium Salt BDBM50303350 Diphosphoric Acid 1-beta-tert-Butyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303346
Diphosphoric Acid 1-beta-tert-Butyl Ester 2-(Uridine-5'-yl)ester,di ammonium Salt BDBM50303346 BDBM222427 5'-deoxy-5'-[4-(β-D-glucopyranosyloxymethyl)-1,2-3-triazol-1-yl]uridine (5)
BDBM222427 5'-deoxy-5'-[4-(β-D-glucopyranosyloxymethyl)-1,2-3-triazol-1-yl]uridine (5) BDBM50199184 CHEMBL214830 5-BROMO-URIDINE-5'-MONOPHOSPHATE 5-bromo-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-dione 5'-monophosphate
BDBM50199184 CHEMBL214830 5-BROMO-URIDINE-5'-MONOPHOSPHATE 5-bromo-1-beta-D-ribofuranosyl(3H)pyrimidine-2,4-dione 5'-monophosphate BDBM50333187 CHEMBL1644891 5'-N-[N-alpha-(2,6-Anhydro-D-glycero-L-gluco-heptit-1-yl-acetamido)-histidinamido]-5'-deoxy-uridine
BDBM50333187 CHEMBL1644891 5'-N-[N-alpha-(2,6-Anhydro-D-glycero-L-gluco-heptit-1-yl-acetamido)-histidinamido]-5'-deoxy-uridine N(4)-Hydroxycytidine Uridine, 4-oxime EIDD-1931 US20240034730, Compound EIDD-1931 Beta-D-N4-hydroxycytidine NHC N4-Hydroxycytidine BDBM430624
N(4)-Hydroxycytidine Uridine, 4-oxime EIDD-1931 US20240034730, Compound EIDD-1931 Beta-D-N4-hydroxycytidine NHC N4-Hydroxycytidine BDBM430624 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-galactopyranoside (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251160 CHEMBL465710
4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-galactopyranoside (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251160 CHEMBL465710 1-(4-(2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl) benzylidene)methoxyamine (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251170 CHEMBL463856
1-(4-(2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl) benzylidene)methoxyamine (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-((methoxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50251170 CHEMBL463856 BDBM50299577 (2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573460
BDBM50299577 (2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573460 {[(2R,3S,4R,5R)-5-(5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27943 uridine derivative, 39
{[(2R,3S,4R,5R)-5-(5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27943 uridine derivative, 39 BDBM50276039 3'-[1-(4-carboxylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL469949 1-[3-(4-CARBOXYPIPERIDIN-1-YL)-3-DEOXY-BETA-D-ARABINOFURANOSYL]PYRIMIDINE-2,4(1H,3H)-DIONE
BDBM50276039 3'-[1-(4-carboxylpiperdinyl)]-3'-deoxy-ara-uridine CHEMBL469949 1-[3-(4-CARBOXYPIPERIDIN-1-YL)-3-DEOXY-BETA-D-ARABINOFURANOSYL]PYRIMIDINE-2,4(1H,3H)-DIONE uridine derivative, 41 BDBM27944 {[(2R,3S,4R,5R)-5-(6-azido-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
uridine derivative, 41 BDBM27944 {[(2R,3S,4R,5R)-5-(6-azido-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid uridine derivative, 42 {[(2R,3S,4R,5R)-5-(6-amino-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27945
uridine derivative, 42 {[(2R,3S,4R,5R)-5-(6-amino-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM27945 uridine derivative, 43 BDBM27946 {[(2R,3S,4R,5R)-5-(6-ethyl-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid
uridine derivative, 43 BDBM27946 {[(2R,3S,4R,5R)-5-(6-ethyl-5-fluoro-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid CHEMBL507060 ((((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(hydroxy)phosphoryl)methylphosphonic acid BDBM50271102 Uridine-5'-alpha,beta-methylene-diphosphate {[(2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-3,4-dihydroxy-tetrahydro-furan-2-ylmethoxy]-hydroxy-phosphorylmethyl}-phosphonic acid 5'-O-[(S)-HYDROXY(PHOSPHONOMETHYL)PHOSPHORYL]URIDINE
CHEMBL507060 ((((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methoxy)(hydroxy)phosphoryl)methylphosphonic acid BDBM50271102 Uridine-5'-alpha,beta-methylene-diphosphate {[(2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-3,4-dihydroxy-tetrahydro-furan-2-ylmethoxy]-hydroxy-phosphorylmethyl}-phosphonic acid 5'-O-[(S)-HYDROXY(PHOSPHONOMETHYL)PHOSPHORYL]URIDINE 5-bromo-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-BROMO-2'-DEOXY URIDINE cid_6035 Bromodeoxyuridine CHEMBL222280 BDBM50207303
5-bromo-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-BROMO-2'-DEOXY URIDINE cid_6035 Bromodeoxyuridine CHEMBL222280 BDBM50207303 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302737 CHEMBL577106
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((hydroxyimino)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302737 CHEMBL577106 6-aza-UMP {[(2R,3S,4R,5R)-5-(3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-2-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid C6-Uridine Derivative, 18 BDBM21340
6-aza-UMP {[(2R,3S,4R,5R)-5-(3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-2-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid C6-Uridine Derivative, 18 BDBM21340 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302745 CHEMBL566863
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-(4-((2-carbamothioylhydrazono)methyl)phenoxy)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50302745 CHEMBL566863 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-6-(4-((methoxyimino)methyl)phenoxy)-2-(3-oxobutyl)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL565507 BDBM50302731
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-6-(4-((methoxyimino)methyl)phenoxy)-2-(3-oxobutyl)tetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL565507 BDBM50302731 Diphosphoric Acid 1''-alpha-D-[1''](4''-Fluoro-4''-Deoxy)-Glucopyranosyl Ester 2-(uridin-5'-yl)ester CHEMBL593125 URIDINE-5'-DIPHOSPHATE-4-DEOXY-4-FLUORO-ALPHA-D-GALACTOSE BDBM50304034
Diphosphoric Acid 1''-alpha-D-[1''](4''-Fluoro-4''-Deoxy)-Glucopyranosyl Ester 2-(uridin-5'-yl)ester CHEMBL593125 URIDINE-5'-DIPHOSPHATE-4-DEOXY-4-FLUORO-ALPHA-D-GALACTOSE BDBM50304034 URIDINE-5'-DIPHOSPHATE-2-DEOXY-2-FLUORO-ALPHA-D-GLUCOSE CHEMBL593830 BDBM50304032 Diphosphoric Acid 1''-alpha-D-[1''](2''-Fluoro-2''-Deoxy)Glucopyranosyl Ester 2-(uridin-5'-yl)ester
URIDINE-5'-DIPHOSPHATE-2-DEOXY-2-FLUORO-ALPHA-D-GLUCOSE CHEMBL593830 BDBM50304032 Diphosphoric Acid 1''-alpha-D-[1''](2''-Fluoro-2''-Deoxy)Glucopyranosyl Ester 2-(uridin-5'-yl)ester JMC521648 Compound 34 {[(2R,3S,4R,5R)-5-(6-cyano-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM21335 6-cyanouridine 5-monophosphate C6-Uridine Derivative, 6
JMC521648 Compound 34 {[(2R,3S,4R,5R)-5-(6-cyano-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}phosphonic acid BDBM21335 6-cyanouridine 5-monophosphate C6-Uridine Derivative, 6 CHEMBL444912 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((1-(4-sulfamoylphenyl)-1H-1,2,3-triazol-4-yl)methylsulfonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50278780 4-(4-{[2',3',4',6'-Tetra-O-acetyl-beta-D-glucopyranosyl]sulfonylmethyl}-1-H-1,2,3-triazol-1-yl)benzenesulfonamide
CHEMBL444912 (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((1-(4-sulfamoylphenyl)-1H-1,2,3-triazol-4-yl)methylsulfonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate BDBM50278780 4-(4-{[2',3',4',6'-Tetra-O-acetyl-beta-D-glucopyranosyl]sulfonylmethyl}-1-H-1,2,3-triazol-1-yl)benzenesulfonamide CHEMBL461514 BDBM50245883 Acetic acid (2S,3R,4S,5R,6R)-4,5-diacetoxy-6-acetoxymethyl-2-(4-formyl-phenoxy)-tetrahydro-pyran-3-yl ester (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-glucopyranoside
CHEMBL461514 BDBM50245883 Acetic acid (2S,3R,4S,5R,6R)-4,5-diacetoxy-6-acetoxymethyl-2-(4-formyl-phenoxy)-tetrahydro-pyran-3-yl ester (2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formylphenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 4-Formylphenyl (2,3,4,6-tetra-O-acetyl)-beta-D-glucopyranoside UTP BDBM50023910 5-azidouridine-5'-triphosphate derivative 1-(3,4-dihydroxy-5-triphosphoryloxymethyltetrahydro-2-furanyl)-1,2,3,4-tetrahydro-2,4-pyrimidinedione Uridine triphosphate (UTP) ({[({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)phosphonic acid
UTP BDBM50023910 5-azidouridine-5'-triphosphate derivative 1-(3,4-dihydroxy-5-triphosphoryloxymethyltetrahydro-2-furanyl)-1,2,3,4-tetrahydro-2,4-pyrimidinedione Uridine triphosphate (UTP) ({[({[5-(2,4-dioxo-1,2,3,4-tetrahydropyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)phosphonic acid BDBM50299576 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573459 (1S)-2,3,6-tri-O-acetyl-1,5-anhydro-1-sulfamoyl-4-O-(2,3,4,6-tetra-O-acetyl-beta-D-galactopyranosyl)-D-glucitol
BDBM50299576 (2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-sulfamoyltetrahydro-2H-pyran-3-yloxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate CHEMBL573459 (1S)-2,3,6-tri-O-acetyl-1,5-anhydro-1-sulfamoyl-4-O-(2,3,4,6-tetra-O-acetyl-beta-D-galactopyranosyl)-D-glucitol BDBM50292723 uridine 2'-phosphate CHEMBL447360 PHOSPHORIC ACID MONO-[2-(2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YL)-4-HYDROXY-5-HYDROXYMETHYL-TETRAHYDRO-FURAN-3-YL] ESTER (2R,3R,4R,5R)-2-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-3-yl dihydrogen phosphate
BDBM50292723 uridine 2'-phosphate CHEMBL447360 PHOSPHORIC ACID MONO-[2-(2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YL)-4-HYDROXY-5-HYDROXYMETHYL-TETRAHYDRO-FURAN-3-YL] ESTER (2R,3R,4R,5R)-2-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-3-yl dihydrogen phosphate 5-fluoro-2'-Deoxy-uridine BDBM50340678 FLOXURIDINE 1-(beta-D-2-deoxy-erythro-pentofuranosyl)-5-fluorouracil 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-fluoro-2'-deoxyuridine 2'-deoxy-5-fluorouridine Fluoruridine Deoxyribose 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione FUDR 5-Fluoro-1-((2R,4S,5R)-4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione 5-Fluoro-1-(4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione CHEMBL917
5-fluoro-2'-Deoxy-uridine BDBM50340678 FLOXURIDINE 1-(beta-D-2-deoxy-erythro-pentofuranosyl)-5-fluorouracil 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)-tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione 5-fluoro-2'-deoxyuridine 2'-deoxy-5-fluorouridine Fluoruridine Deoxyribose 5-fluoro-1-((2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione FUDR 5-Fluoro-1-((2R,4S,5R)-4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione 5-Fluoro-1-(4-hydroxy-5-hydroxymethyl-tetrahydro-furan-2-yl)-1H-pyrimidine-2,4-dione CHEMBL917 (bradykinin triacetate)2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid (BK) H-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg-OH CHEMBL406291 bradykinin BDBM50049949 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid(Bradykinin)
(bradykinin triacetate)2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid (BK) H-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe-Arg-OH CHEMBL406291 bradykinin BDBM50049949 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid 2-(2-{[1-(2-{2-[2-({1-[1-(2-Amino-5-guanidino-pentanoyl)-pyrrolidine-2-carbonyl]-pyrrolidine-2-carbonyl}-amino)-acetylamino]-3-phenyl-propionylamino}-3-hydroxy-propionyl)-pyrrolidine-2-carbonyl]-amino}-3-phenyl-propionylamino)-5-guanidino-pentanoic acid(Bradykinin)